Benzyltrimethylammonium hydroxide did not arrive in labs through some grand announcement; curiosity about phase-transfer catalysts and quaternary ammonium compounds in the mid-1900s opened the door. Before the 1960s, many chemists relied on harsh conditions to drive reactions involving ionic species. Someone figured out that swapping sodium or potassium ions out for larger, organic ammoniums could make mixing oily reactants with water-based ones a whole lot easier. Among these, benzyltrimethylammonium hydroxide became a quiet champion. Its historical roots stretch through organic synthesis, where researchers sought gentler conditions and higher yields. Over time, this compound grew popular with chemists running organic reactions that once demanded plenty of patience and even more brute force.
You can walk into a chemical warehouse or scroll through catalogs and find benzyltrimethylammonium hydroxide, usually as a 40% solution in water or methanol. It lands in your hands as a colorless to pale yellow liquid, promising strong basicity and a whiff of something ammoniacal. Researchers, industrial chemists, and manufacturers don’t treat it as just another salt. Its power to shuttle ions between phases makes a tough reaction a bit more bearable. The role isn’t always glamorous, but it anchors synthesis labs that value reliability.
What hits you first is the strong odor. It's not the kind of stuff you want to sniff, but that sharpness signals a potent base. The solution remains stable, though it'll darken if left out or exposed to contaminants. It shows basicity levels that rival sodium hydroxide, and unlike many solids, it avoids clumping or caking. With a melting point too high to touch and a boiling point that doesn't matter in typical lab settings, its practical importance shines in solution. Water and alcohols act as perfect partners, dissolving the salt so thoroughly that nothing gets left behind. In most applications, its high ionic strength and surface activity win over those searching for a stubbornly effective reagent.
You see numbers like "40% solution in water" on the label. That’s not an arbitrary figure—manufacturers stick to this concentration because it balances storage safety and ease of use. Lesser concentrations risk dilution errors, stronger ones add handling hazards. Quality suppliers go out of their way to specify trace impurities, water content, and recommended storage temperatures. SDS sheets call out its strong basicity and the sensation it can cause if spilled or splashed. Transport labels flag its corrosive nature and the requirement for tight-sealing caps. Many labs train their people to recognize the warning signs and keep dedicated containers ready for waste disposal.
Making benzyltrimethylammonium hydroxide usually starts with its chloride salt, produced from benzyl chloride and trimethylamine. The chloride gets treated with aqueous silver oxide or ion-exchange resins to swap out the chloride ion for a hydroxide. The final solution’s purity gets confirmed by titration; the unmistakable sharp smell and intense reaction with acids leave no room for doubt. The process is simple on paper, but quality matters, because any leftover chloride or foreign ions can sabotage delicate syntheses.
This compound’s core talent is as a phase-transfer catalyst, pushing reactions between folks who'd rather ignore each other—aqueous and organic reactants. In practice, it can help peg halides off alkanes, pull stubborn protons from organic acids, and promote alkylations in non-polar media. When you need to tweak the molecule, swapping the benzyl or methyl groups gives access to new reactivities or solubilities. Its strong basicity supports various condensations or eliminations, and it happily hands over its hydroxide ion in reaction after reaction without forcing the issue.
On a shelf you might see “Triton B,” “BTMAH,” or “benzyltrimethylammonium hydroxide.” Chemical catalogs repeat the same few aliases, but anyone with time in academic labs will point out Triton B as the colloquial favorite. Sometimes it shows up under trademarked labels from big chemical houses, but the formula stays consistent. Multiple identifiers allow folks from different continents and backgrounds to find common ground on this staple.
Skin and eye contact with benzyltrimethylammonium hydroxide brings pain fast. Lab safety briefings need to highlight splash risks, as this compound can leave chemical burns or respiratory distress. Proper gloves, tight goggles, and fume hoods aren’t optional here; they’re the difference between an ordinary day and a bad one. In spills, neutralization with acids works, but not before isolating the affected zone and warning others in the area. Storing the solution away from acids and oxidizers avoids surprise reactions. Disposal must respect local regulations—this is not a “down the drain” situation. With solid procedures, it stands out as manageable rather than menacing.
Its most loyal followers work in organic synthesis, especially for reactions involving phase transfer or strong bases. I’ve seen it drive Williamson ether syntheses, support the generation of ylides, and simplify alkylations that seemed impossible under standard aqueous conditions. More recently, it found its way into analytical chemistry, helping to release specific ions and streamline sample preparation. Environmental chemists rely on it for decomposing rigid matrices or breaking down organics. Pharmaceutical researchers and fine chemical manufacturers appreciate its predictability in multi-step syntheses, and the electronics sector chooses it to etch silicon wafers with precision. Wherever ions feel unwelcome or hard to move, benzyltrimethylammonium hydroxide can step in.
The world of chemical synthesis never stops looking for improvements, and benzyltrimethylammonium hydroxide serves as a baseline catalyst. Research labs continue to examine its behavior with new classes of substrates, hoping to drive up yields or cut down on by-products. Smart tweaks to its structure spark new discussions each year, with patents popping up for more efficient, less hazardous analogs. Customizing conditions to cut energy costs or speed up slow conversions fuels plenty of PhD projects. Progress sometimes means stripping out solvents, recycling the catalyst, or switching to greener production routes. Those shifts speak to a demand for practicality balanced with environmental impact.
Toxicology studies flag this quaternary ammonium compound as caustic and potentially irritating above certain concentrations. Skin and eye irritation dominate the risk profile; inhalation is only a concern during glovebox mishaps or open transfers without ventilation. Chronic toxicity studies in animals remain scarce, but acute exposures demand fast action. Occupational health guidelines stress education, quick shower access, and regular PPE audits. Most labs track near-misses and update their safety protocols yearly. In all these years working in shared lab spaces, I’ve seen the lesson repeated—nobody wants to be the reason the eyewash station gets a workout.
Organic synthesis rides on the back of innovation, but some tools keep returning for good reason. For all its simplicity, benzyltrimethylammonium hydroxide stays in favor because of its reliability and adaptability. Future research could hinge on modifying its structure to reduce toxicity or increase efficiency. Efforts to blend it with greener solvents or recycle solutions give it another edge. Scaling up industrial production demands optimizations for waste minimization and energy use, which means more jobs for process engineers and safety professionals. Collaboration between academic labs and manufacturing floors might land new versions with fewer hazards and better selectivity, stretching a familiar compound’s reach even further into tomorrow’s chemistry toolkit.
Walk through any laboratory or talk to folks in chemical manufacturing, and Benzyltrimethylammonium Hydroxide, better known as Triton B, crops up more than you’d think. This compound acts as a strong base that teams up with other chemicals to help them change or react faster. Over the years, I’ve seen chemists reach for it not just out of convenience, but because it gets things done where weaker bases won’t cut it.
This substance finds plenty of work as a phase-transfer catalyst. Basically, it moves stuff from one place to another in a chemical mixture, letting things that don’t want to mix on their own finally shake hands. In real terms, this means companies use it to make medicines, plastics, dyes, and coatings. In research settings, it helps scientists speed up or improve reactions that build new molecules or change old ones. From routine experiments to large-scale production, Triton B sticks around because it pulls off tough tasks consistently.
It’s easy to overlook chemistry’s role in products we use each day. The paints that brighten up a room, the electronic gadgets that run smoothly, detergents that clean out stains, and even tablets that fight off a headache — each involves some careful chemistry. Benzyltrimethylammonium Hydroxide often works behind the scenes to make these products possible. Think of the number of plastics and coatings in everyday items, and chances are, somewhere in their production trail, Triton B had a hand in the process.
The strengths of Triton B don’t come without costs. Its caustic nature can mean serious risks if not respected. I’ve watched experienced chemists double-check gloves, goggles, and have extra rinsing stations on hand before popping open a bottle. As much as it helps industries move forward, its use brings demands for responsibility. In the wrong hands, or without training, it can cause burns or harm the environment if spilled or dumped carelessly. Regulations make sure storage stays secure and disposal doesn’t end up hurting people or wildlife, but compliance still depends on attention to detail at every step.
With its usefulness clear, the next question becomes one of balance: How can we meet our chemical needs without risking health or polluting communities? Some research groups have started exploring greener catalysts that sidestep the harsher impacts of traditional ones like Triton B. These alternatives sometimes draw from plant material, or use recyclable supports to cut down on waste. Change takes time, and switching up a tried and true factory process isn’t easy. Yet, demand for products that don’t put people or the planet on the line keeps nudging industry leaders and scientists toward solutions that fit both safety and productivity.
The more we push for innovation, the more important it gets to ask what tools we’re relying on in laboratories and factories. Benzyltrimethylammonium Hydroxide delivers real results. As more people pay attention to the footprint of everyday goods, it’s up to chemists, companies, and lawmakers to set high standards, work together, and keep searching for better answers. Everybody wants products that work, but nobody benefits if progress comes at the price of health and future security. Making smart choices about what we use in the lab and factory floor shapes not just what we buy — but how we live.
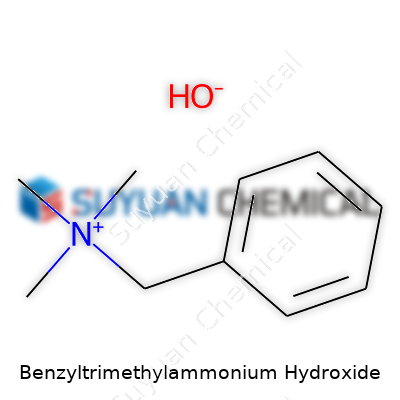
Benzyltrimethylammonium hydroxide, known to many chemists as Triton B, pops up often in both research and industrial chemistry. The formula is C10H17NO. That brings together a benzyl group, a nitrogen surrounded by three methyl groups, and a hydroxide ion. This configuration matters for more than just academic curiosity; it shapes how the molecule behaves in real-world applications.
Misreading or mistyping a formula can do more than ruin an experiment. In some roles, I’ve seen incorrect formulas send projects sideways for weeks or even months. A mixup of even a single atom can lead to entirely different compounds and outcomes. Working in synthesis labs, we often relied on the right formulas to avoid wasted time, reduce costs, and, more importantly, prevent dangerous reactions. It sounds simple, but double-checking chemical formulas feels like a low-tech safety net in a high-tech field.
Benzyltrimethylammonium hydroxide earns its stripes as a strong organic base. The molecule's structure gives it the punch that scientists look for, especially during phase-transfer catalysis and in organic synthesis. It can help move ions between water and organic solvents, allowing for reactions that wouldn't work otherwise. In my own bench work, especially preparing cyclizations and alkylations, I saw how it let us tackle reactions cleanly, with fewer side-products. From pharmaceutical ingredients to the creation of custom polymers, the reach of this compound runs broad.
Sitting in the chemical store-room, Benzyltrimethylammonium hydroxide comes in concentrated solutions, which means splashes and fumes can hurt skin, eyes, and lungs. I've watched what happens when new lab workers skip safety data sheets; it only takes one careless moment to learn respect for caustic chemicals. Knowledge and attention help keep accidents at bay. Proper labeling and accessible material safety data help, as does regular safety training and transparent communication about storage and handling requirements.
Communication about chemical compounds still shows cracks. Digital databases can hold outdated or wrong information, so cross-checking with peer-reviewed literature and manufacturer data remains critical. Publication of errors and near-misses, especially with quaternary ammonium compounds, would help the community avoid repeating mistakes. Regular audits—the kind I once dreaded but later valued—also force teams to revisit old assumptions and update protocols.
People handling chemicals make decisions every day with broad implications. Correct chemical identification, paired with vigilance about storage and reactivity, protects more than just the experiment. In my experience, a culture that expects curiosity, question-asking, and ongoing learning creates safer labs and better science. The chemical formula of Benzyltrimethylammonium hydroxide might not change, but the way we use and share knowledge should keep evolving for everyone’s benefit.
People who spend time in research labs or chemical manufacturing sometimes run into names like Benzyltrimethylammonium Hydroxide—usually called TMAH for short. The stuff pops up in various industrial jobs: making pharmaceuticals, handling organic syntheses, and even in electronics production. As someone who’s stood in a lab and handled it, I know the slick, soapy feel of TMAH and the way its fumes can cling to the back of your nose. The question that crops up again and again: just how hazardous is TMAH?
One look at the safety data sheets, and it’s clear: this is not your everyday cleaning product. TMAH can cause some nasty burns, and it’s notorious for attacking both skin and eyes. Just a splash can leave raw red welts, raising the risk of long-term scarring. Eyes don’t fare any better—there’s a genuine chance of permanent damage if splashed, with government agencies setting strict first-aid protocols. Inhalation is rough too; its vapor irritates airways, causing coughing, choking, and in bad cases, fluid buildup in the lungs. TMAH has been involved in fatal accidents, especially in factories making semiconductors in Asia. The smallest contact with just a few milliliters of concentrated liquid can become deadly without rapid medical treatment.
TMAH’s danger doesn’t stop with burns. Its toxicity actually comes from its effect on the human nervous system. If someone absorbs enough—either through skin, swallowing, or breathing—the substance messes with nerve signaling. Muscles can seize up or turn floppy, breathing falters, and heart rhythms get knocked off course. This isn’t just theory; published reports from Asia have documented cases where an unsuspecting worker suffered cardiac arrest after a spill.
In my time with chemical safety officers, everyone treated TMAH with the same seriousness as old-school dangers like cyanide or hydrofluoric acid. Some companies require two people present for every handling session and demand triple redundancy in protective gear. No shortcuts.
The solution doesn’t revolve around banning TMAH entirely—it has industrial uses nobody can easily replace. The answer lies in tight precautions: splashproof goggles, chemical resistant gloves, and face shields at a bare minimum. Training makes a difference, not just for lab staff but also for janitors and drivers who transport the stuff. It helps to have written emergency procedures on hand: showers, eyewash stations, and plenty of ventilated air.
More companies could switch to less concentrated forms where possible, or automate tasks so fewer people come into contact with TMAH. In the few years since learning just how dangerous this stuff gets, I noticed some plants borrowing safety ideas from high-risk settings—using sensors, specially designed containers, and even robotics for the dirtiest jobs.
Some scientists have started exploring safer alternatives, but so far, nothing does exactly what TMAH does in semiconductor cleaning. Until there is a true replacement, everyone who comes close to it benefits from vigilance, education, and a work culture that doesn’t allow shortcuts. Chemical safety isn’t glamorous, but it sure beats the alternative.
Benzyltrimethylammonium hydroxide plays a role in research labs and industry settings alike. On the safety data sheets, it stands out for its strong basic properties and its tendency to react with air and moisture. This makes it a bit unpredictable if left unchecked. Years spent in university labs have taught me that even small mistakes in how one stores bases like this have created messes nobody wants to fix. Besides, nobody wants a chemical burn or a corroded shelf. That risk alone should set off alarm bells.
Direct sunlight, warm rooms, and fluctuating humidity all spell trouble for benzyltrimethylammonium hydroxide. Exposure to any of these speeds up decomposition, and once that process starts, not only does the chemical lose effectiveness—you also face potential leaks and pressure buildup. I’ve seen dedicated students surprised when a bottle cap bulged; that wasn’t just wasted product but a sign of higher risks in the storage room. The solution sits in clear practices: keep it in tightly sealed containers, glass if possible, and always away from acids and metal surfaces that might trigger violent reactions.
A crowded chemical cabinet leads to confusion. Sharply distinct labeling shouldn’t become optional. In one shared university lab, a simple colored sticker system helped prevent accidental mixing—an approach that’s both cheap and easy to follow. Store benzyltrimethylammonium hydroxide far from strong acids, oxidizers, or anything flammable. If space runs out, don’t cut corners by doubling up; use a secondary containment tray. This catches leaks before you start mopping up a caustic spill from the floor and helps keep the workspace clean and safe. A transparent logbook or digital records help teams stay on the same page about storage dates and container checks.
Chemical storerooms should stay cool and dry. An ordinary fridge proves tempting, but direct contact with food risks contamination. A dedicated chemical refrigerator, kept between two and eight degrees Celsius, works best. Outside the fridge, placing containers on lower shelves limits the chance they’ll tumble or break open. Good airflow matters in case vapors escape; a vented chemical cabinet, not just a locked closet, gives staff a line of defense. Forgotten storage corners build up risks over time because air can stagnate and concentrate fumes. A regular inspection schedule lays eyes on aging labels, damaged closures, or other warning signs.
No chemical storage plan survives without people who understand what’s at stake. Every new person joining my lab got a run-through on strong base storage. We’d practice finding the nearest eyewash and spill kit. Chemical burns from caustics like benzyltrimethylammonium hydroxide stick in your memory longer than safety lectures ever could. Keeping a good supply of gloves, splash goggles, and chemical-resistant aprons right by the storage area sets a clear expectation for anyone handling the bottle. Emergency numbers near the storage location finish the picture. The habits you build here translate directly to safer labs—and fewer close calls.
Trust grows in labs that build habits around safety. No fancy equipment needed—just a shelf, a label, and the will to follow a checklist every week. Even outside of compliance requirements, a shared sense of responsibility keeps everyone sharp. Chemical incidents rarely come from not knowing the rules—they come from not caring. Benzyltrimethylammonium hydroxide doesn’t forgive mistakes, and neither should your storage plan.
Benzyltrimethylammonium hydroxide shows up in labs mostly as an aqueous solution. It’s rarely sold pure because the solid form can get unstable and hard to manage. The most common bottle you’ll find carries a 40% solution, usually in water, sometimes in methanol. Other popular concentrations include 20% and 25%. Lower concentrations, like 10% or 15%, show up if the process asks for more diluted alkali or if the user wants finer control in a tricky reaction.
Chemists building organic molecules often reach for the 40% option. I used it while running a phase-transfer reaction in graduate school and remember how the bottle needed to be tightly sealed and handled with care, since leaks could make a whole bench sticky and smelly. The higher strength packs more punch, so you use less volume for the same effect, which helps in scaling up or keeping water content low.
For most research, reagent grade does the trick. This grade is made with high-purity expectations and works for academic or industrial synthesis. Analytical grade is even cleaner, but not as easy to find because most applications don’t need it. For reactions that demand less trace-metal content or fewer unwanted ions, a specialty high-purity grade comes into play.
Commercial supply catalogs present these as “reagent,” sometimes “puriss,” or just by the stated percentage in water or methanol. The labeling isn’t always crystal clear, so the best move is to check the certificate of analysis for each lot. I’ve learned that two bottles labeled “40% in water” might behave differently if one batch came with higher chloride or heavy metal traces.
Some folks might wonder why the details matter. In organic synthesis, the smallest impurity or the wrong water content can change a yield, introduce side products, or make purification harder. A strong base like this will change pH or break apart delicate functional groups in seconds, so knowing the actual strength saves a lot of headaches later. In catalysis, certain metals or ions from lower grades can poison a catalyst and tank an expensive reaction.
In the lab, I’ve handled 25% and 40% grades and learned fast that the higher one will aggressively eat through some plastics and destroy gloves that aren’t meant for it. I saw a colleague lose a morning because he thought he was using 20% and grabbed a 40% solution instead. The results? He over-alkylated his product and had to restart from square one.
To avoid trouble, check the label every time and consult the supplier’s documentation. Keep incompatible chemicals far from storage and use proper protective gear. Use a chemical fume hood every time. Double-check reaction protocols to match the required grade and concentration to the recipe. During purchasing, choose suppliers who provide a batch-specific certificate of analysis, not just generic specs. Swap advice with colleagues or check literature for recommended suppliers—peer experience can save hours of unnecessary troubleshooting.
For scale-up and industry use, invest in regular training for handling caustic bases and keep emergency eyewash stations nearby. Consider dilution steps only with distilled water and in controlled conditions. When possible, start small to watch for unexpected results before going full-scale. Benzyltrimethylammonium hydroxide brings plenty of power to chemistry, but working with the right concentration and grade keeps the process smoother and a whole lot safer.