Dioctyldimethylammonium bromide found its place in chemistry labs as quaternary ammonium compounds started gaining real traction in the twentieth century. Laboratories searched for antimicrobial solutions during global difficulties like World War II, and these compounds showed promise as germicidal agents. The demand for surface-active agents shaped their story. Over decades, advances in organic synthesis enabled formulation tweaks, influencing commercial applications from water treatment to personal care. Understanding this background shows how shifts in health, hygiene, and industry have long driven developments in chemical manufacturing.
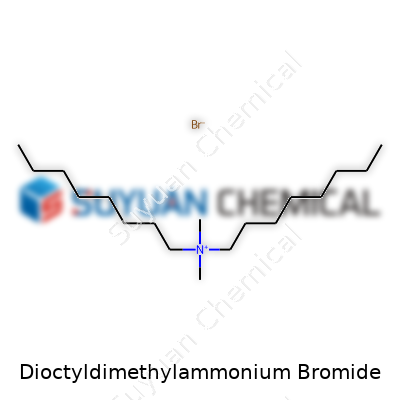
People interested in specialty chemicals notice Dioctyldimethylammonium bromide for its robust surfactant properties. Found as a white, crystalline solid, this compound’s oil-loving octyl chains give it a unique place among quats. It dissolves well in both alcohols and water, making it flexible for applications where both fat solubility and water compatibility matter. This versatility has made it attractive to formulators in areas like disinfectants, textile processing, and even phase transfer catalysis. Its chemical formula—C18H40BrN—packs both potency and adaptability into a single product, changing the routine of many working chemists and plant engineers from generation to generation.
For those mixing solutions, Dioctyldimethylammonium bromide demonstrates reliability, melting around 49–54 °C and resisting rapid breakdown across a broad pH spectrum. Unlike some more delicate surfactants, it tolerates hard water without dropping out or losing power. From my own lab days, consistency means peace of mind; there’s less fiddling with conditions or worrying about variable sources. Its relatively stable structure also keeps it from quickly decomposing under storage, as long as humidity and temperature don’t go out of control.
What matters most on a barrel label? Purity, moisture content, and free amine—all checked to fit both regulatory standards and high-performance demands. Many suppliers guarantee greater than 98% purity, minimal water content, and clear identification codes such as CAS number 5538-94-3. Transporters and handlers know they’re dealing with a substance requiring standard chemical product labeling: hazard identification, storage guidance, and clear warning icons for safe handling in transit and in use.
Dioctyldimethylammonium bromide starts its journey by reacting dioctylamine with methyl bromide and a suitable base. The synthesis, while straightforward for the trained chemist, needs careful attention to temperature, reaction time, and by-product management. Purification—often achieved by recrystallization—becomes key for both efficiency and compliance, keeping unwanted reactants or side products at bay. Watching bench-scale batches react, seeing the crystals form, and understanding each step’s purpose gives a level of appreciation that goes beyond a datasheet. Each tweak in process can mean smoother production or a reduction in costly waste.
Chemists who want to modify the core quaternary structure have explored swapping out counterions or attaching functional groups for special purposes. By replacing bromide with chloride, or introducing hydrophilic or bulky groups, it's possible to tune solubility, antimicrobial action, or compatibility with unusual polymers. Dioctyldimethylammonium bromide holds its structure in both acidic and slightly basic environments, so it serves well in multi-component chemical reactions or composite materials where other additives might fail or break down. Its reactivity remains modest under everyday conditions, a big plus for anyone seeking predictability and stability.
Searching across technical catalogs, this compound turns up under names like DODAB, dioctyldimethylammonium bromide, and a collection of supplier trade names meant to catch attention without causing confusion. For those accustomed to short-hand jargon, “DODAB” sticks in memory and conversation. Cross-referencing specifications becomes crucial in global trade—seeing how each region, company, or regulatory agency might list the same substance in slightly different ways.
Strict rules surround Dioctyldimethylammonium bromide’s use, handling, and disposal. Anyone who’s loaded drums or poured out a beaker in a lab coat knows to keep layers between skin and substance: gloves, goggles, plenty of ventilation. Contact may bring irritation, and higher concentrations trigger more acute effects. Safety Data Sheets call for immediate first aid and robust spill control procedures. In industrial scale storage, secondary containment and temperature controls keep inventories safe for workers and the environment. Plant workers keep a trained eye on exposure limits, trusting both training and clear labeling to reduce risk.
Day to day, Dioctyldimethylammonium bromide ends up in more places than most realize; from stabilizing cement slurries in construction, to biocidal components keeping hospital surfaces germ-free. Water treatment plants rely on its ability to rupture microbial membranes, while laundry detergents use it to enhance softness and kill odor-causing bacteria. In my experience, research teams also lean on it for liposome formation—helping carry drugs across cellular barriers in drug delivery models. That wide reach means supply stability and sourcing ethics stretch into multiple sectors at once.
Laboratories keep pushing forward by investigating modifications to DODAB for better performance in new roles, such as reducing toxicity, improving biodegradability, or increasing compatibility with green chemistry. Synthetic tweaking and advances in formulation continue to drive its evolution. Academic and commercial researchers share findings in journals and patents, prompting manufacturers to adjust recipes and tweak manufacturing “on the fly.” I recall projects tweaking chain lengths or swapping for greener solvents, as customer demand shifts as fast as new studies arrive.
Toxicological studies point out real risks at both high doses and in chronic exposure scenarios. Skin and eye irritation come up repeatedly, and aquatic toxicity puts rules in place for wastewater discharge. Researchers have looked into bioaccumulation, with some studies showing it breaks down slowly and may build up in sensitive environments. Regulatory agencies keep a close watch on new toxicology data, often prompting updates to hazard classifications and use restrictions, especially in areas close to drinking water supplies. Those working with the substance in labs and plants learn to respect both acute risks and the slow trickle of long-term hazards.
Looking ahead, expectations for cleaner, safer, and more efficient chemicals are already changing the way Dioctyldimethylammonium bromide will be produced and used. Demand for low-toxicity antimicrobials, along with stricter environmental standards, will push companies to adapt formulation methods and invest in bio-based raw materials. More research is aimed at recycling or degrading quats after use, opening new doors for sustainable lifecycle management. Consumers and regulators alike expect full transparency on chemical sourcing, manufacturing footprint, and downstream effects—which pushes the whole industry to think past short-term sales and towards lasting solutions. As labs and companies keep looking for alternatives or improved formulations, new generations of DODAB can address both legacy applications and emerging needs.
Factories, labs, and cleaning supply chains rely on all sorts of niche chemicals to keep their operations moving. Dioctyldimethylammonium bromide is one of those names that doesn’t roll off the tongue, but it pops up in more places than most people recognize. Its strong antimicrobial action explains why many cleaning products, industrial sanitizers, and even specialty coatings list it among their ingredients.
In daily life, most don’t give much thought to how disinfectants manage to kill bacteria and fight off mold. With dioctyldimethylammonium bromide, the science works in its favor. It belongs to a chemical group called quaternary ammonium compounds. These compounds disrupt microbial cell membranes, sending bacteria and fungi into retreat. Across hospitals, food processing equipment, and public restrooms, this compound’s power shows up in spray bottles, wipes, and surface-safe liquids. Hospitals count on products with this compound to reduce the risk of healthcare-associated infections, which affect millions worldwide each year.
The usefulness of this chemical stretches even further. Manufacturers often add it to paints, coatings, and plastics to keep surfaces free from mold and algae, especially in damp environments. Think of HVAC coils in large buildings or the walls of indoor swimming pools. It finds a spot in the treatment of cooling towers and industrial water systems, keeping biofilm and bacteria from gumming up the works.
It doesn’t end there. In oil and gas drilling, some fluids turn slimy because of microbial growth—this compound is used to prevent that. Chemists lean on its antistatic qualities when blending antistatic agents for fuel and lubricants.
Every time a disinfectant product wipes out surface germs, there’s a chance that improper use or disposal leaves trace chemicals behind. Not every home cleaner with advanced antimicrobials ends up in the landfill or water system, but enough of these chemicals work their way into streams to give researchers pause. Studies in Europe and North America highlight the effect of quaternary ammonium compounds building up in soil and water, affecting aquatic life.
The worries don’t stop at the environment. Like other strong disinfectants, direct skin exposure or accidental inhalation causes issues for sensitive people. Skin irritation, allergic reactions, and respiratory distress can follow misuse. Safety data sheets recommend gloves and good airflow around the area. In commercial agriculture, guidelines require workers to wear masks and goggles during large-scale application.
People need solutions that hit germs hard in clinics, restaurants, schools, and on factory floors. But using these potent chemicals wisely means looking beyond the label. Smart storage, careful application, and responsible disposal matter. Alternatives do exist—some products now use hydrogen peroxide or alcohol-based sanitizers instead, especially in places where chemical buildup threatens wildlife or water supplies. Regular public education, clear packaging, and stronger regulations all give consumers and professionals better tools for safe use.
At the intersection of health, safety, and environmental impact, dioctyldimethylammonium bromide shows just how much chemistry shapes daily routines—and why an ounce of caution goes further than a gallon of cleaner.
Dioctyldimethylammonium bromide shows up in cleaning products, disinfectants, and some industrial processes. People might not see it in their kitchen cupboards, but factories, hospitals, and labs work with it all the time. This chemical does a good job as a disinfectant, which makes it popular in settings where killing bacteria and viruses matters. So, it’s not a household name, but it’s not rare either.
Studies and workplace safety reports suggest this chemical gives people trouble when they inhale it, touch it, or get it in their eyes. It acts as an irritant. Workers report red, itchy skin, stinging eyes, sore throats, or coughing fits. Long-term contact without decent protective gear might make these problems last longer or turn worse. The National Institute for Occupational Safety and Health (NIOSH) and other agencies flag it for occupational health reasons.
I once spent a summer at a factory that made cleaning supplies. Even small splashes caused some coworkers to break out in rashes. The safety sheets got passed around with plenty of stories about red hands and watery eyes. Nobody forgot their gloves after that. When I look at those experiences, it’s clear: ignoring safety around strong cleaning chemicals invites health trouble.
Prolonged or repeated skin contact can turn into dermatitis. Some reports link misuse with aggravated asthma, especially for people who already faced breathing problems. Research points to similar chemicals in the quaternary ammonium family causing allergic reactions for susceptible folks. Animal tests with related compounds revealed possible impacts on liver function after long-term, high-dose exposure. For regular consumers, the risk seems small, but those handling large amounts all day can face real issues.
Regulators pay close attention. The Environmental Protection Agency calls for clear labeling and expects products with it to include full handling instructions. Safety Data Sheets (SDS) list Dioctyldimethylammonium Bromide as harmful if swallowed, inhaled, or placed in contact with skin without protection. In the EU, the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) regulation screens its use. The difference between safe use and harm comes down to training, protection, and following guidance from those agencies.
Businesses can cut down health problems by giving workers gloves, eyewear, and face masks. Training sessions help people learn where this chemical lurks and how to avoid spills or inhaling fumes. Good air ventilation takes away some danger in laboratory and industrial sites. I always found that regular reviews of safety procedures prevented people from getting complacent. The same goes for reading product labels at home and storing containers out of reach of children.
Industry can keep pushing for safer alternatives, but for now, responsibility falls on each user to read safety labels, use equipment right, and not skip personal protection. Experience and data both point to a simple truth: respect for chemical hazards is what keeps people healthy.
Dioctyldimethylammonium bromide shows up in plenty of lab and industrial spaces. It's one of those specialty chemicals often used as an antiseptic, disinfectant, or surfactant. From handling inventory in research labs to supporting daily routines in production environments, safely working with chemicals like this ranks high on the to-do list. Neglecting storage guidelines doesn’t just cause paperwork headaches; it can risk health, wreck batches, and even violate safety codes. The right approach keeps the work moving without unwanted surprises.
Humidity harms dioctyldimethylammonium bromide over time. It’s a hygroscopic chemical, meaning it absorbs water straight from the air. Once moisture creeps into containers, purity drops. Wet granules start clumping and can break down, which threatens accuracy in both research and production. Heat ramps up the problem, speeding up degradation and sometimes triggering unwanted chemical reactions. For this reason, storing it in a cool, dry spot matters. Stash it away from any direct sunlight, radiators, or heat sources. Climate-controlled rooms or dry cabinets work well to keep the chemical stable.
Hands-on work with chemicals has taught me that even reliable packaging breaks down if not closed properly. Dioctyldimethylammonium bromide comes in tightly sealed drums, jars, or poly bags, and those seals aren’t just for shipping. Every time a container opens, the risk for moisture and contaminants rises. Dust and grime don’t just look bad—they can introduce substances that interfere with the chemical’s function and safety. Screw on caps right away. Use clean scoops for each transfer. Label containers with dates to make rotation easy, so the oldest supply gets used first.
Mixing chemicals accidentally causes more emergencies than most folks imagine. Dioctyldimethylammonium bromide shouldn’t share shelf space with strong oxidizers, acids, or reducing agents. Oxidizers could spark a hazardous reaction, while acids and other aggressive chemicals break down the compound or churn out dangerous byproducts. Sorting shelves by chemical class and keeping a well-marked inventory list goes a long way. If possible, keep it in a dedicated cabinet away from reactive products.
Clear labeling helps during busy periods or staff changes. Use bold, readable labels including product name, handling warnings, and storage date. Leaks and spills happen, so it pays to keep spill control material within arm’s reach. That means having gloves, absorbent pads, and a step-by-step cleanup guide right near storage. Treat any messes with urgency; skins and eyes react sharply on contact.
As someone who’s helped onboard newcomers, I’ve seen how focused safety training changes routines for the better. Regular refreshers make sure everyone recognizes the risks and storage details unique to dioctyldimethylammonium bromide. Having an up-to-date Safety Data Sheet on hand means answers are never far away. Written procedures for storage and handling help keep everyone on the same page, not just during audits, but every day.
Stable storage keeps dioctyldimethylammonium bromide doing its job without wasted stock, surprise downtime, or safety scares. Not only does this save money, it protects workers and the integrity of scientific, medical, and industrial results. The daily habit of checking lids, watching temperature and humidity, and staying organized pays off in reliability and peace of mind.
Dioctyldimethylammonium bromide, often shortened to DODAB, brings together some straightforward chemistry and tangible real-world uses. This compound features a central nitrogen atom with two methyl groups and two octyl chains attached, balanced by a bromide ion hanging on the side. Structurally, the nitrogen holds a positive charge, attracting bromide as its partner. Each octyl group is simply a long, eight-carbon chain.
Chemically, the formula comes out as C18H40BrN. Draw it out, and you’ll see a nitrogen core with arms splaying out—two short, stubby methyls up top, two lengthy octyl arms swinging low, and a bromide counterion nearby. This configuration makes the molecule act like a double-tailed surfactant. In simple terms, one side feels friendly toward water, and the longer chains make it easier to slide into layers or membranes.
This structure—bulky, lopsided, and, in a sense, both water-loving and fat-loving—helps DODAB fit into places most other molecules can’t. Drop it in water, and it starts sticking itself together into little round clusters called micelles. That “sticky” nature gives DODAB a spot in industrial cleaners, sanitizers, and fabric softeners. Hospitals rely on it because those long octyl chains can bust apart bacterial cells, making surfaces safer.
Years ago, working in a research lab, I learned how fine molecular tweaks can tip the scales between a substance being a lab curiosity or a market staple. Counting on just four big hydrocarbon chains or something shorter changes everything—water solubility, toxicity, and even cost. DODAB gets the balance right, especially when paired with bromide, which makes it more water-soluble than the chloride version sometimes used in similar settings.
I always pay attention to the safety sheet; DODAB, like other quaternary ammonium compounds, won’t win a “gentlest chemical of the year” award. High doses can irritate skin or lungs, and reckless disposal doesn’t mix well with water systems. Quats build up in rivers and lakes unless treatment plants pull them out efficiently.
Some European regulations have tightened rules against overuse, especially for substances that pile up in the environment. Testing shows long carbon chains resist breaking down in nature, which means informed handling matters more than marketing promises. Not everything safe on your shirt stays safe in your ecosystem. Being mindful of rinse water, minimizing extra use, and following disposal advice can limit environmental downsides.
Companies developing new formulations know DODAB gets results, but reputations shift along with research. Long-term safety studies and investment in greener breakdown options—especially enzyme-based “digesters”—help. Looking for alternatives, like shorter-chain quats or switching to biodegradable surfactants where possible, makes a big difference.
Chemical structure isn’t just a formula on paper. It means real-world impact in every bottle and bucket. That double-octyl, double-methyl blend makes DODAB a workhorse compound, but responsibility never stops at the factory gate. By understanding its shape and its stubbornness, both as a cleaner and as a residue, we all take part in shaping cleaner, safer environments.
Nobody likes to think about dangerous chemicals building up around them, but this is a problem that hits close to home for anyone working with industrial compounds or running a lab. Dioctyldimethylammonium bromide isn’t a household name, but its uses stretch from disinfectants to surfactants. Because of its antimicrobial properties, it ends up in places like hospitals and cleaning solutions—environments already juggling safety. The last thing anyone wants is this substance finding its way into local waterways or landfill sites, causing headaches for communities and the environment.
From experience in research labs, I know firsthand how small spills or careless storage can put people at risk. Dioctyldimethylammonium bromide can irritate the skin, eyes, and even the respiratory system. Even more troubling, its long-term ecological impact is tough to untangle if it enters soil or water, harming aquatic life and, in turn, the food chain. Though not as headline-grabbing as oil spills, improper handling can snowball over time.
Many would prefer to just toss old chemical containers in the garbage or drain, but that runs against strong scientific evidence and clear-cut environmental regulations. The way forward calls for careful and planned steps:
I’ve seen situations where shortcuts led to workers falling sick or small fires breaking out because someone underestimated how a chemical like this could behave near heat or sunlight. Safety instructions matter, but so does a culture where asking questions about chemical disposal feels encouraged, not brushed off as someone being too cautious.
Regular training goes a long way towards keeping bad habits from taking root. Having procedures in writing helps, but periodic practice where everyone demonstrates the process cements the knowledge and highlights areas that might trip people up in real life.
Every time someone safely disposes of dioctyldimethylammonium bromide, they help keep drains and soil free from contamination. Local water sources stay clean, aquatic ecosystems remain healthy, and communities avoid long-term headaches. Proper handling of this chemical isn’t difficult, but it does ask for a small investment of effort and the wisdom to rely on specialists when needed.