Before anyone started calling it by its technical name, N,N-Dimethyldecanamide had humble beginnings in the world of organic chemistry. Back in the mid-twentieth century, chemists pushed for new compounds that blended stability with versatility—qualities often sought for both lab research and industry. Like many amides before it, its creation stemmed from curiosity about how tweaking molecular chains could change behavior. Early patents targeted improvements in lubrication and surfactancy, bringing it traction in manufacturing and agricultural sectors. Over decades, increased demand for specialized solvents meant further refinement, better sourcing of raw materials, and tweaks inspired by both trial and regulatory pushback. Today, the story of N,N-Dimethyldecanamide traces a path driven by necessity and adaptation, echoing the long-standing human urge to make advanced materials safer and more efficient.
At its core, this compound is an amide formed by combining decanoic acid derivatives and dimethylamine. With a clear or pale-yellow appearance, it manages to slip into all sorts of processes without drawing much attention. What makes it valuable isn't flash or notoriety—it's the way it solves practical problems by acting as a solvent, plasticizer, or even a stabilizer under demanding conditions. Its appeal has less to do with any one “wow” feature and more with quiet persistence: fitting seamlessly in diverse product formulations for agriculture, pharmaceuticals, and cleaning agents. When I think of how it operates, the analogy of a backstage hand in theater comes to mind—vital, often overlooked, and consistently reliable.
N,N-Dimethyldecanamide falls under the category of long-chain fatty amides, shaped by an extended hydrocarbon tail attached to a central amide group. It has a boiling point close to 310°C, allowing it to withstand high temperatures before decomposing. The chemical doesn’t dissolve well in water, but it meshes smoothly with organic solvents such as ethanol, diethyl ether, and chloroform. Its density hovers around 0.880-0.900 g/cm3 at 25°C, and its consistency veers from oily liquid to waxy solid, depending on the climate and purity. The low vapor pressure keeps emissions in check, which certainly comes in handy in enclosed worksites. Having handled similar chemicals, I know firsthand how these characteristics allow operators to control exposure and waste without huge investments in new equipment.
Manufacturers ship products with clear technical datasheets specifying assay purity—usually above 98% for high-grade batches. Labels list the IUPAC name, CAS number (108-83-8), UN codes for safe transport, and handling notes per OSHA’s Hazard Communication Standard in the U.S. Specifications highlight refractive index, color, residual water content, and acid/base numbers. These details sound dry until you’ve worked in production, where clarity on impurity levels translates directly to reduced process downtime and fewer rejected lots. I’ve seen how small lapses on labeling can set off protracted investigations, so precision in documentation stands out as more than just compliance hoop-jumping.
Industrial synthesis follows a straightforward path: reacting decanoyl chloride with dimethylamine under controlled cooling, usually in the presence of a base like pyridine. The chemical reaction—acylation—kicks off with a strong release of heat, so plant operators monitor closely to avoid runaway temperatures. The byproduct, hydrochloric acid, gets neutralized or scrubbed in parallel, diverting waste streams safely. Post-reaction, purification happens through distillation or crystallization to reach target purity. There’s no perfect route; experienced chemists look for yield or byproduct risk and adjust temperatures or solvents accordingly. I’ve seen enthusiasm for “greener” methods grow, especially with pressure from environmental regulation. Some teams experiment with milder conditions or alternative solvents, although scale-up often reveals hidden compromises.
A defining feature is chemical resilience—no surprise since amides sit low on the reactivity spectrum. With enough heat or strong acid/base, the amide bond gives way, splitting back into carboxylic acids and amines. Likewise, this backbone allows for subtle structural tweaks, either by lengthening the alkyl chain or swapping out the amine group. These adjustments unlock changes in solubility, melting point, and compatibility with other chemicals. Some researchers modify it further for specialty use in pharmaceuticals or as an intermediate in the synthesis of ionic liquids. From my own experience with related amides, custom-modification projects spur both innovation and patience. One small change in functional group often ripples through entire product lines, setting off new rounds of safety and regulatory review.
Industry shorthand for N,N-Dimethyldecanamide isn’t exactly catchy, but it fills its role. It pops up as DMDA, Decanamide, N,N-dimethyl-, and sometimes Decanoic acid dimethylamide. Trade names vary by manufacturer, each carving out minor branding territory for marketing or inventory purposes. Keeping up with these alternate names—especially in multinational operations—calls for clarity in documentation. Even a small paperwork mix-up due to synonyms can lead to confusion across borders, something I’ve seen stall shipments or spark unnecessary audits more than once.
No matter how benign a substance looks on paper, real-world safety shapes its reputation. For N,N-Dimethyldecanamide, standards stretch beyond labeling. Workers dealing directly with it wear gloves and goggles, especially if they’re exposed to heated tanks or aerosolized mists. The chemical irritates eyes and skin with prolonged contact, so most plants keep showers and eyewash stations close at hand. Safety Data Sheets (SDS) walk through emergency procedures and storage rules: keep containers cool, label clearly, and avoid sparks or flames. Knowing how fast a routine job can slip into emergency mode, I’ve seen management teams drill on spill responses and lockouts for this family of chemicals, aiming to reduce panic and injuries.
N,N-Dimethyldecanamide finds itself at the intersection of convenience and necessity. In agriculture, it acts as a solvent for pesticide formulations, helping active ingredients dissolve and spread through crops more evenly without breaking down under sun and moisture. In industrial cleaning, it dissolves tough greases and supports stable emulsions in heavy-duty detergents. Plastics and resins benefit from its presence as a plasticizer, imparting flexibility and process reliability. Other uses trickle into cosmetics, personal care products, and pharmaceutical manufacturing, often quietly, as an excipient or processing aid. Having spent time in both agricultural and manufacturing environments, I’ve seen how it transforms unruly mixtures into stable, usable products, saving costs on both raw materials and troubleshooting.
Innovation never really rests, especially for chemicals straddling multiple industries. Research teams lean on N,N-Dimethyldecanamide when formulating safer, lower-toxicity pesticide carriers or developing specialty solvents with minimal environmental footprint. Ongoing work investigates modifications at the molecular level—how branching or alternative amines affect degradation rates and compatibility with new actives. Lab-scale testing often points the way to improved resistance to outdoor degradation, which can mean longer-lasting agricultural effects with less runoff. Pharmaceutical trials explore whether its incorporation as a solubilizer unlocks new drug delivery profiles. Across every project, the heavy focus on user and environmental safety keeps scientists iterating, balancing efficiency with real-world constraints. It’s not uncommon to see a spike in patents whenever regulations tighten or new classes of active ingredients emerge.
A chemical’s reputation rises or falls with its safety profile. For N,N-Dimethyldecanamide, acute toxicity for humans and animals appears low. Most studies find only mild to moderate irritation in skin or eyes with ordinary contact. Inhalation hazards stay limited by virtue of low volatility, although the use of PPE in manufacturing keeps exposure to a minimum. Chronic toxicity and environmental fate, though, still draw close scrutiny. Data gaps exist regarding long-term breakdown in soil or water, raising questions about environmental persistence or accumulation in food chains. Regulatory authorities, like the EPA and European Chemicals Agency, regularly call for updated research, with testing focused on biodegradation, aquatic toxicity, and potential for bioaccumulation. From a practical standpoint, being upfront about unknowns—and tracking research progress—channels both trust and the right investments, which I find builds stronger partnerships between producers and regulators.
Looking forward, the future of N,N-Dimethyldecanamide depends on its continued relevance as a customizable, relatively safe building block. The ongoing shift toward greener chemistry stands as both a challenge and an opportunity. Developers invest in bio-based sourcing and closed-loop production to minimize ecological impact. Regulatory shifts steer industry toward substances with full toxicological transparency, rewarding manufacturers that invest in lifecycle assessments and alternative testing. Advances in molecular design, supported by data analytics and high-throughput screening, could steer the next generation of related amides toward even lower hazard profiles or specialty performance niches. As someone who’s seen enthusiasm surge and fade on the shop floor and in research labs, I know sustainable momentum only holds when everyone—from plant worker to policymaker—learns from both the successes and shortfalls of chemicals like N,N-Dimethyldecanamide. Environmental, social, and governance criteria keep shaping its path forward, making it a compelling case study in the real-world dance between innovation, safety, and responsibility.
N,N-Dimethyldecanamide turns up in more places than most people would think. For most folks who have never heard the name, it sounds like something meant only for lab coats and safety goggles. In reality, this compound touches many corners of modern life. It shows up quietly in products under the sink, in fields outside town, and even inside the food chain.
This amide forms a backbone for agriculture. Farmers need help fighting pests that nibble away at crops or lay waste to their yields. N,N-Dimethyldecanamide acts as a carrier for pesticides. Its structure lets active ingredients dissolve easier and get delivered straight to where the bugs live or feed. When working with crops, every percentage point of efficiency counts. By boosting how well pesticides blend and coat plants, this compound helps farmers use less to get better results. Less spraying saves fuel, labor, and ends up with fewer chemicals running off into water systems.
Look under the kitchen sink and you might find legacy products enhanced by this ingredient. The amide travels into floor strippers, degreasers, and other cleaning agents. Its main gift lies in its ability to soften and break up grime, grease, and oil. Professional cleaning calls for something powerful but safe to handle, and this compound finds a middle ground. It lets cleaning formulas lift out the tough stuff without leaving irritating residues. In my time working in a restaurant kitchen, products using amide-based surfactants always beat the more basic options. Food prep spaces stayed clear of grease, and we didn’t end up with harsh fumes filling the back room.
N,N-Dimethyldecanamide shows strength as a solvent, especially in paints and coatings. Manufacturers lean on it to thin out products so they apply smoothly on surfaces. Some industrial coatings—especially those used for heavy machinery or large construction gear—rely on this type of solvent because it keeps the paint workable but doesn’t evaporate too quickly or cause the finish to sag. For folks in maintenance or renovation, this small detail makes repainting less of a gamble. The surface comes out even, tough enough to avoid chips, and doesn’t peel off months down the line.
Clothes have to feel right and last through many washes; textile processing uses N,N-Dimethyldecanamide to help with dyeing and softening. Its ability to let dyes seep into fibers means colors stay bright, and the clothes hold up better. On the personal care front, formulators slip this compound into creams or lotions for that soft, silky glide most people want in a moisturizer. Companies aim to blend oil and water in ways the skin can absorb, and here the amide acts as a sort of behind-the-scenes peacemaker for different ingredients.
It’s not all smooth sailing. As with many chemical helpers, safety comes down to how thoughtfully people handle and dispose of it. Manufacturers have to keep worker safety in mind and look at new green chemistry routes when possible. Looking ahead, more eco-friendly substitutes and tighter regulation will probably shape where this compound appears. Until then, soaking up its benefits calls for a careful eye on how it touches people, crops, and the environment.
N,N-Dimethyldecanamide shows up in more places than most realize. Chemical companies often use it as a solvent or carrier in pesticides, coatings, and specialty industrial fluids. In my own work, I’ve seen it on ingredient lists and safety datasheets enough times to pay attention. The question of whether this substance can be handled safely isn’t just an academic exercise. Workers in agriculture and manufacturing deal with it up close, and communities live near places where it gets stored and processed.
It’s smart to look at facts first. Available studies show that N,N-Dimethyldecanamide has relatively low acute toxicity through skin or inhalation. It’s not a known carcinogen. Still, that’s not a free pass. Prolonged contact can irritate skin and eyes. Inhalation at high concentrations can bother the respiratory tract, lead to headaches, or even cause drowsiness. Some animal research also hints at liver and kidney effects from repeated exposure at high doses. Anyone using it should take potential for harm seriously.
I’ve chatted with industrial hygienists who point to one constant: chemical safety always leans on process, not assumptions. Even though this solvent doesn’t make the top of the hazard list, it can still cause real problems, especially if basic rules get ignored. Workers need gloves, goggles, and decent ventilation, even if the job seems routine. I recall a plant manager sharing a story where a minor spill led to eye irritation for several employees—nobody wants to relive that. Supervisors set the tone by reinforcing responsible behavior and easy access to material safety data sheets (MSDS).
Once it leaves the drum, N,N-Dimethyldecanamide enters the environment one way or another. Some laboratory results show low toxicity for fish or aquatic invertebrates. Yet the lack of long-term studies on persistence and breakdown means that full impacts remain unclear. Disposing of leftover material or wash water down regular drains can send the chemical to municipal systems not equipped to filter it out. I’ve worked on site audits in regions where waste mismanagement led to unexpected contamination—no one thinks it’ll happen until it does.
Real safety springs from a mix of good habits and good information. Proper labeling, storage in sealed containers, and regular training all help reduce risk. I’ve seen companies arrange annual refreshers on chemical hazards—sometimes dry, always important. Keeping emergency showers and eyewash stations maintained is not just a checkbox but a practical backup. And for smaller workshops or independent farmers, reaching out for expert advice from poison control or state agencies can make a huge difference.
Calls for more transparent data on chronic health effects aren’t going away. While government agencies and researchers have done some testing on N,N-Dimethyldecanamide, industry insiders know the gaps in knowledge. Until there’s more clarity, anyone working hands-on with this chemical ought to keep up strong safety habits and treat every product update or safety bulletin as a priority, not an afterthought.
Trust grows between employees and employers with every day that passes without a mishap, not because a chemical promises to be “safe” but because everyone takes steps to look out for each other. My experience shows that people who ask hard questions about chemicals—and act on what they learn—help build a culture that values long-term wellbeing over shortcuts.
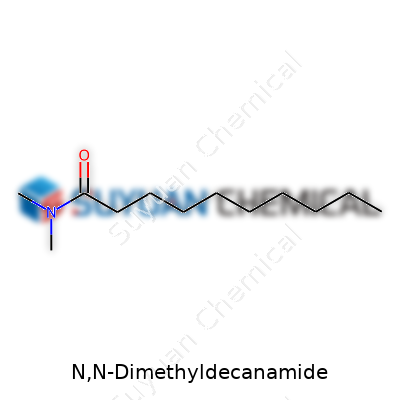
Chemical Formula: C12H25NO
Molecular Weight: 199.33 g/mol
N,N-Dimethyldecanamide carries the backbone of a fatty acid amide. At its core, this compound forms by taking decanoic acid and swapping out its carboxylic hydrogen with a dimethylamine group. The result is a straight, twelve-carbon chain with two methyl groups attached to the nitrogen. What looks simple on paper—twelve carbons, twenty-five hydrogens, one nitrogen, and one oxygen—opens up more possibility than its formula lets on.
The length of the decyl chain means this amide loves showing up where hydrophobic behavior matters. It holds its own in organic synthesis, lubricants, and in processes that hinge on both oil affinity and moderate polarity at the head. Its melting behavior and solubility shift based on the balance between non-polar tail and polar head. Chemists count on it when they want to harness that balance—breaking up stubborn mixtures, acting as a solvent, or softening plastics. The fact that the formula points to a middleweight molecule—neither light enough to fly away, nor so heavy it gums up works—explains why industries look to this compound for compatibility.
Practical chemistry always means more than theory. Carelessness with any compound, even one as seemingly docile as N,N-Dimethyldecanamide, presents risk. Some amides can irritate skin or cause health concerns. They call for gloves, ventilation, and clear labeling so nobody grabs a container without a clue what’s inside. Policies stressing clear hazard communication in the workplace matter more than ever. A safety data sheet pinned in the storage room beats memory every time.
Environmental impact also can’t get left out. Once upon a time, solvents ended up down the drain and out of mind. Now stricter wastewater rules ask everyone to think twice about where speciality chemicals end up. Biodegradability, toxicity to aquatic life, and persistence—these factors urge us to keep track of every step from delivery to disposal. Some amides resist breaking down, so partnering with waste handlers who understand what’s at stake keeps regulators off your back and communities safe. That makes accurate labeling not just a legal requirement, but a moral one.
In real labs, formulas matter, but people matter more. Training and regular review become the difference between clean records and costly incidents. Teams who make time for cross-training know why clear procedures prevent small spills from becoming big disasters. Stockrooms that use barcoding and inventory management quickly spot leaks before they snowball into emergencies. Transparency lets newcomers ask questions when something smells off or doesn’t add up.
Looking across the supply chain, more firms choose greener synthesis paths and work to eliminate legacy chemicals from routine processes. Substituting safer alternatives wherever possible drives innovation. It pushes for continuous updates on evolving industry guidance, so today's practical choice does not turn into tomorrow’s regulatory headache.
Chemists, safety officers, and handlers share responsibility for using even the most ordinary-sounding compounds wisely. That means knowing the formula—C12H25NO—and its story, but also making decisions grounded in facts, data, and personal accountability. Lessons from campus labs up to global producers shape habits around safety, stewardship, and respect for detail. In the end, every measured decision—labeling, storage, substitution—shows commitment not just to productivity, but to a profession built on trust and expertise.
Not every chemical sparks curiosity, but spend enough time in any chemistry lab and you start to realize that routine creates safety. N,N-Dimethyldecanamide, with its growing role in industrial and agricultural spaces, deserves that level of care. Over my years talking to lab managers and plant workers, one thing stands out—cutting corners in chemical storage never pays off. Even with compounds like this one, which aren’t usually high on the hazard scale, mistakes still come with consequences.
This substance acts as a solvent in a range of uses, from pesticide formulation to specialty coatings. It doesn’t ignite as quickly as some other solvents, but don’t let that lull you. Many chemicals only pose a problem once someone gets casual about the basics—temperature, humidity, and containment. When liquid solvents leak or vaporize, even in small amounts, workers notice headaches, skin irritation, or even eye problems. The risk multiplies in spaces with poor airflow or when multiple chemicals interact.
Start by keeping it in containers that don’t react—stainless steel or high-quality plastic drums work well. Metal that corrodes or breaks down leads to leaks, which spike cleanup costs and product loss. Sealing the cap after every use sounds simple, but I’ve watched spills happen because people took shortcuts on little things.
Cool, dry, and shaded conditions matter. Insulation from direct sunlight or heat sources prevents unexpected decomposition. I’ve seen warehouses run into trouble because excess heat warped containers or sent fumes into the air systems. Keep temperature as steady as possible, aiming for around room temperature. Humidity control helps as well, given that moisture creeping into bottles adds an unnecessary layer of risk.
Store N,N-Dimethyldecanamide away from oxidizers and acids. Even infrequent contact can trigger reactions, especially with concentrated solutions. Speaking from experience, chemical compatibility charts shouldn’t sit forgotten in a drawer—they guide storage plans more than most people think. Keep those printed and visible near storage racks.
Clear, detailed labeling wins every time. Include the chemical name, hazard warnings, and the date received. Rotating stock from oldest to newest might not sound exciting, but old materials break down, which increases your risk. I’ve seen students grab the wrong bottle because faded labels were never replaced. Those small mistakes snowball under pressure.
Lockable storage cabinets, preferably ventilated, create another barrier against spills or unauthorized access. Teenagers in schools or new workers in factories both need extra protection. Having a key system or swipe cards adds accountability to the mix—a smart move for any organization with high staff turnover.
Even with careful storage, accidents will happen. That’s why spill kits, gloves, and eyewash stations deserve a spot nearby. Regular training helps staff respond quickly instead of panicking. OSHA and EU-REACH standards both push for accessible Safety Data Sheets (SDS), and rightfully so. I’ve watched a first-year tech in a panic nearly make things worse simply because she hadn’t reviewed the emergency protocols for the chemicals in her care.
The best chemical storage routines develop from paying attention to past mishaps and sharing those lessons. N,N-Dimethyldecanamide might not draw the kinds of headlines that other chemicals do, but storing it wrong leads to real, preventable problems. A disciplined approach backed by real-world advice, readable labels, and accessible safety tools makes all the difference between business as usual and a costly incident.
Every stage of the chemical supply chain brings a different challenge, and shipping N,N-Dimethyldecanamide shows why caution and practical decision-making are worth more than a checklist. Over the years, I’ve seen that packaging isn’t just a formality—it's fundamental to safety and solid business. N,N-Dimethyldecanamide serves as a useful solvent across multiple industries, but the rules for moving it from manufacturer to end user don’t budge. Reliable transport and the right packaging keep both people and the environment safe.
Companies rely on two typical container types: tight-head steel drums and high-density polyethylene (HDPE) drums, often sealed with tamper-evident lids. I’ve handled shipments in both kinds, and the container you choose depends as much on regulatory compliance as your risk tolerance. HDPE drums hold up against corrosive spill risks and often work for bulk orders. Steel drums deliver extra durability for longer hauls or rough terrain in transit. Smaller quantities sometimes go into jerricans or properly rated bottles, each sealed to minimize evaporation and accidental exposure. No matter the container, clear safety labeling is not optional—workers need transparency about what they’re dealing with.
Nobody wins by cutting corners with statutes or ignoring the fine print. The UN assigns N,N-Dimethyldecanamide a code to identify its hazards, and shipment depends on matching transport regulations for road, air, and sea. From experience, improper paperwork or a misclassified package brings shipments to a grinding halt. Worse, inadequate packaging can result in leaks, fires, or environmental contamination, leading to huge fines or legal action. I strongly recommend working with logistics partners who show they understand chemical shipping and offer real references, not vague claims.
Plastic pollution and hazardous waste show up in newsfeeds for a reason. I’ve seen manufacturers offer recyclable drum return programs, cutting down on landfill impact and helping companies meet their own green targets. Many buyers look for suppliers committed to minimizing waste, using multi-use drums, or offering take-back recycling. It doesn’t just feel good—it helps with public relations and, more importantly, aligns with tightening national rules on solvent waste. Give preference to suppliers who share a recycling strategy and can show their process audits.
Insurance and tracking emerged as the real backbone for accountability. Some companies now tag drum shipments with QR codes for instant traceability and safety checks at every stop. Over-the-road carriers often request SDS documents—Safety Data Sheets—on hand each time, giving emergency responders clear information if an accident occurs. Training staff to follow these safety procedures, from sealing caps to stacking drums, prevents minor errors from snowballing into disasters.
From my years working alongside warehousing teams and compliance officers, the smartest companies set up review sessions after each shipping cycle. They check for leakage, update shipping logs, and cycle out damaged drums. Embracing this review culture sharply drops incident rates. Future-focused companies now experiment with eco-packaging and digital shipment monitoring, aiming to shrink environmental impact and increase efficiency at once.
Investing up front in the right shipping materials and partners is how professionals in the chemical sector keep their edge. Packaging and transport for N,N-Dimethyldecanamide, done right, builds trust, protects people, and proves commitment to responsible supply chain management.