Chemical innovation rides on the coattails of curiosity and necessity. N,N-Dimethyloctyldecanamide earned its stripes as a response to global calls for improved surfactants by the late 20th century. As industries tried to move away from legacy solvents that left heavy environmental footprints, this amide-based compound provided a balance between performance and reduced toxicity. Academics in applied chemistry wanted alternatives to N,N-dimethylformamide and older fatty acid amides. Factories and formulation labs moved steadily, incorporating new sources of fatty chains from plant oils, while synthesizing higher molecular weight N,N-dialkylamides. Supply improved, and demand shifted. Over the decades, N,N-Dimethyloctyldecanamide got its due as an ingredient that truly earned a spot in technical handbooks and agricultural compendiums.
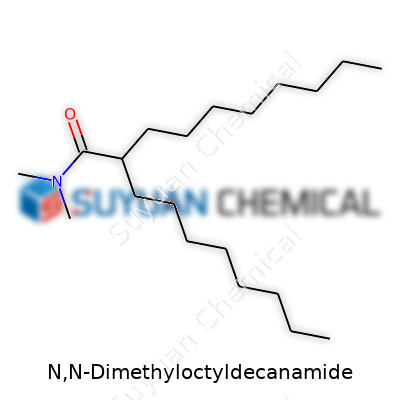
This compound forms part of the dialkylamide family. Manufacturers describe its molecular structure as a blend of hydrophobic and hydrophilic units, supporting both solubility and emulsification. With a mix of an octyl group, a decanoyl backbone, and two methyl groups linked to the amide nitrogen, it delivers more than a sum of its parts. In daily industry work, this molecular layout matters, helping people push the boundaries in pesticide mixing, polymer dispersions, and as a solvent. Knowing what each part of the structure does leads formulators like me to push its utility further.
As a clear to pale yellow liquid around room temperature, N,N-Dimethyloctyldecanamide displays a faint amine scent and a mildly viscous flow. Its melting point usually falls below 0°C, which means no handling issues in cooler warehouses. Boiling points hover above 300°C, keeping it thermally stable in a range of applications. Density sticks near 0.87–0.92 g/cm³, not heavier than standard water-based systems—making pump adjustments at the plant a non-issue. The compound resists hydrolysis under most pH conditions. As a non-ionic surfactant, it behaves differently from cationic or anionic agents, which often brings it to the front for tasks needing gentle but persistent emulsification. It dissolves well in hydrocarbons and alcohols, and it remains inert enough to play nicely with active ingredients in tank mixes.
Clean labeling counts for plenty, especially as regulations grow tighter over time. Lot certificates usually detail purity above 95%, with GC-MS confirming precise peaks for N,N-Dimethyloctyldecanamide. Water content needs to stay below 0.5%, keeping hydrolytic breakdown at bay. Many technical data sheets warn about trace amine or acid by-products, urging buyers to check for faint alkaline residues before large-scale use. Drum labels carry hazard codes for skin and eye irritation, and safety data sheets chart flash points above 185°C. Each drum or pail must also mention batch traceability, which supply chain managers and regulatory auditors lean on heavily, especially in Europe and the US. Specific labeling for REACH compliance lines up with trending discussions about exposure and low-level chronic toxicity.
The typical route starts with fatty acid chlorination—octyl or decanoic acid first hits thionyl chloride or phosphorus trichloride to create the acid chloride, and this then runs with dimethylamine under vigorous stirring. This acid-amine condensation forms the amide linkage and yields N,N-Dimethyloctyldecanamide, sometimes with a catalytic base pulling up final yields. Because of the volatility of dimethylamine and risks linked to acid chlorides, site engineers need closed reactors and scrubbers to avoid venting hazardous gases. Those who have spent time supervising these syntheses recognize the importance of triple-checking temperature controls. The post-reaction blend gets washed, neutralized, and dried through vacuum stripping—at this point, plant chemists keep a sharp eye on color and odor before approving drumming. Recrystallization rarely fits these viscous liquids, so fractionation helps polish out most side products.
The backbone of N,N-Dimethyloctyldecanamide resists many attacks, but strong acids or bases at elevated temperatures will cleave the amide bond, reverting it to its constituent fatty acids or amines. In rare cases, peroxides or ozone can oxidize terminal alkyl groups, shifting polarity and solubility. A few creative chemists have tried esterification of the residual carboxamide or alkyl chain halogenation to introduce new bioactivities, though the irony is that the pure, unmodified amide already brings plenty to the table for current users. Cross-linking with other amides or epoxides remains more theoretical, but research groups sometimes publish work on copolymerizing amide groups with certain acrylates, chasing improvements in soil release and cleaning formulations. Actual adoption in commercial settings depends on cost, ease of use, and final product approval.
Industry catalogs often list N,N-Dimethyloctyldecanamide under a half-dozen names. Some call it Octyldecyl dimethylamide, a nod to the two main carbon chains. A handful of agri-chemical suppliers use trade names that lean on the “fatty amide” label for better shelf recognition, though these don’t differ much in terms of molecular content. Chemists grew up memorizing IUPAC names, while suppliers and formulators keep a running log of commercial monikers across North America, Europe, and Asia. This pile of synonyms sometimes leads to confusion on import forms, so clarity counts—customs or safety authorities want every detail spelled out, from CAS number to full chemical formula.
Handling practices don’t just prevent accidents—they protect livelihoods and health long after the workday. N,N-Dimethyloctyldecanamide doesn’t vaporize or burn like acetone, but contact sensitization leaves unlucky workers itching and red. Full nitrile gloves protect operators during transfer and batching, and splash goggles block rare but nasty eye contact. Many regional regulations ask companies to keep spill kits on hand, with adsorbent socks and neutralizers ready. Fire protection standards require foam or dry agent extinguishers for spaces storing bulk chemical stocks. Workplace air monitoring for amine odors reassures operators that closed systems really work. Years of plant experience taught me that quick safety refreshers make a difference—especially as seasonal staff cycle in or when production ramps push capacity. Regulatory audits, near-miss reporting, and proper PPE keep most facilities running trouble-free, saving headaches that come with missteps.
Formulators in agriculture grabbed onto this molecule for its drift reduction and adjuvant roles, especially in pesticide sprays. In textile processing, its softness and antistatic behavior found homes in finishing agents. I’ve seen cosmetic manufacturers use it as a gentle co-emulsifier. Water treatment specialists value it for helping stubborn oils drop out. Environmental scientists sometimes add it to oil spill response blends to aid hydrocarbon dispersion in open water—a job that standard solvents never quite mastered without environmental drawbacks. Cleaning product designers tweak their recipes for better removal of greasy soil with this amide. The ride through industry always circles back to one lesson: task-specific use beats one-size-fits-all.
Innovation isn’t quiet in the world of amide surfactants. Research labs, university departments, and private firms explore its performance in biodegradable blends, seeking lower aquatic toxicity without losing surface tension or solubility. Teams try pairing N,N-Dimethyloctyldecanamide with biosurfactants from yeast or fermentation broths to tighten its green reputation. Oilfield researchers track its performance under high-shear drilling environments, and they measure foaming, solvency, and lubrication in side-by-side tests with classic detergents. Academic chemists sometimes publish novel routes that skip hazardous acid chlorides, leveraging enzyme-catalyzed amidation for greater safety—these approaches wait on scale-up cost drops. Lab life for these compounds always blends a sense of trial-and-error with the reality of production economics and regulatory signoff.
Public health always brings the conversation back to chronic exposure and acute risks. Toxicologists running tests on N,N-Dimethyloctyldecanamide usually see low acute toxicity by oral or dermal routes, with LD50 numbers much higher than volatile solvents. Researchers probe both aquatic and human cell lines for chronic risk profiles. Occupational health practitioners look at skin sensitization and inhalation studies, noting irritation at higher levels but little evidence for carcinogenicity or mutagenicity. Environmental scientists check how much seeps into surface water, watching for low bioaccumulation—a huge step up from older solvent systems. Direct exposure above allowed thresholds triggers local inflammation, so process managers install proper extraction or automate heavy transfers. Policy groups require robust toxicology files before approving fresh uses, so the world’s safety database for this amide keeps growing each year.
Watchful eyes in the specialty surfactant markets signal more growth for N,N-Dimethyloctyldecanamide as new application spaces open up. Producers hover over trends around sustainable chemicals, shaping supply by shifting to feedstocks from non-edible plant oils. Research into enhanced oil recovery, biodegradable cleaning products, and renewable crop protection points to a growing footprint for this compound. Regulatory landscapes keep moving, with Europe and North America tightening limits on persistent organic pollutants and older amide solvents, leaving space for this less-hazardous option. Product managers and plant technologists continue asking for safer, greener performance, which nudges both developmental and operational work. My own reading of market signals makes it clear—tailored chemistry and well-documented safety stories open doors for compounds like N,N-Dimethyloctyldecanamide, especially as industry and society both demand cleaner, smarter ingredients at scale.
Sometimes the names in chemical ingredient lists leave people scratching their heads. N,N-Dimethyloctyldecanamide, for example, doesn’t roll off the tongue. Still, for anyone paying attention to what’s in household products, this compound pops up more than you might think. It’s a nonionic surfactant, which in plain language means it helps mix oil and water. In my experience organizing the cleaning supplies under my kitchen sink, most labels list a cocktail of ingredients designed for this very job: keeping things stable, breaking through grease, or helping other compounds stay dissolved.
Looking closer, this substance plays a strong supporting role in liquid laundry detergents, household cleaners, and some crop protection chemicals. Its main skill lies in boosting the cleaning power of products. Grease and grime come off faster, and the stains rubbing shoulders with the fibers of my favorite jeans practically lift away on their own. In agriculture, it helps pesticides and fertilizers move evenly across plant leaves, so farmers don’t end up with patches of missed coverage or runoff pooling at the edges of fields.
Folks paying attention to ingredient lists may feel uneasy seeing chemicals they can’t pronounce. I share that gut reaction, especially as more people look for “clean” or eco-friendly products. N,N-Dimethyloctyldecanamide stands out because, compared to many surfactants, it breaks down more easily in the environment. According to reports from the European Chemicals Agency, it doesn’t persist as much as older surfactants. That matters to rivers, lakes, and the wildlife living there. Cleaning routines at home rarely cross my mind as having downstream effects, but surfactants often wash out to municipal water systems or backyards, so how long they linger does matter.
Studies from regulatory groups find low risk to people when products use it as directed. In cleaning products or in the field, most exposure happens in small amounts. Still, those working in factories or mixing spray tanks have a bigger stake in safe usage, so protective equipment and good labeling stay important. Risk doesn’t disappear just because something goes to market, and ongoing reviews help check for new evidence about health or environmental risks. If you work with chemicals or feel responsible for what goes down the drain, full transparency from manufacturers and regulators helps sift hype from fact.
Whenever I talk to folks about buying greener or safer products, I encourage checking ingredient transparency. More brands now list what’s inside and offer contact info for deeper questions. For N,N-Dimethyloctyldecanamide, focus lands on sourcing and biodegradability. Companies that use renewable plant-based sources or share biodegradation data win more trust. Responsible manufacturing keeps worker safety and local air quality in sight. For those in agriculture, training workers and relying on integrated pest management can lessen heavy chemical reliance.
As consumers, supporting companies that disclose more and push for smarter chemistry makes a real difference. Choices at the store add up, nudging big brands to fine-tune their ingredient lists and seek better alternatives. If anything, my experience sorting through cleaning products taught me to read the small print, ask questions, and back up my worries with solid info. At the end of the day, knowing a bit more about compounds like N,N-Dimethyloctyldecanamide helps us all scrub a little smarter.
N,N-Dimethyloctyldecanamide, sometimes called by trade names or as a fatty acid amide in ingredients lists, pops up in shampoos, conditioners, and shower gels. Chemists use it to thicken liquids and help surfactants mix. Few shoppers notice it listed near the bottom of lengthy labels, but the conversation about its safety keeps growing among those who examine what goes on their skin.
Most of the evidence about N,N-Dimethyloctyldecanamide’s safety comes from toxicological studies completed by suppliers and published by independent researchers. The Cosmetic Ingredient Review (CIR) has assessed this ingredient in the past, pointing out that it doesn't show acute toxicity at common use levels in skin or eye tests. Its structure is similar to other fatty acid amides that people have safely used for many years.
At the same time, the industry sees no clear sign of skin irritation or allergic reaction in humans outside rare circumstances. That lines up with government agency assessments—both European and North American regulators have not flagged it for harm at the concentrations found in shampoo or soap. The Environmental Working Group lists it as low-risk for health concerns based on available research.
Reading and trusting the ingredient list becomes tricky because new chemicals show up before long-term data can speak for itself. Many people skipped over synthetic names like this one in the past, but interest in “clean beauty” is pushing brands to source better studies and find safer alternatives for any doubtful ingredient.
From years of covering personal care product science, I’ve seen trends: Moms, teenagers, and folks with sensitive skin lead the push for simple formulas. With N,N-Dimethyloctyldecanamide, even though most evidence points to safety, shoppers who deal with contact dermatitis or eczema might want to talk with a dermatologist before adding something new to their routine. Anyone noticing redness or irritation after a new product would do well to double-check labels and test patch, since sensitivities can vary.
Another side worth mentioning: The ingredient usually shows up in small percentages—just enough to make products feel smoother and easier to rinse away. Regulatory agencies set strict rules for how much can be in a given product, so the chances of overexposure through standard use look slim.
Where demand goes, product development follows. If shoppers show broad concern about any ingredient, stores and brands tend to hunt for plant-based, biodegradable swaps. Research in this space moves faster than ever, especially as pressure mounts over environmental impact and microplastic pollution. I’ve talked with formulators who say that as clean label trends grow, synthetic chemicals—safe or not—get replaced with naturally sourced alternatives to please cautious customers.
People with allergies, young kids, or health conditions might want to lean into minimal formulas or shop from brands with transparent third-party testing. The science points to low risk with N,N-Dimethyloctyldecanamide as used in personal care today. Still, the call for more independent, long-term research persists. Until then, educated choices and open conversations with healthcare professionals will help keep shelves both innovative and safe.
N,N-Dimethyloctyldecanamide doesn’t come up in daily conversation, but its use matters in cleaning, agriculture, and plenty of manufacturing processes. This compound mixes an amide group with two long carbon chains—think of it as a hybrid that brings flexibility and power, especially in surfactants and detergents. What stands out is its ability to lower the surface tension of water. That means dirt and oil don’t put up much of a fight once this stuff hits the scene.
You probably care less about molecular diagrams and more about real-world performance. In practice, this compound isn’t afraid of greasy messes. The structure lets it dissolve oils and fats most soaps struggle with. Once, cleaning up fryer grease with regular dish soap left me scrubbing for ages. A specialized cleaner listing this exact amide on the label cut the time in half and left less residue. The science behind that comes down to how those extra-long chains trap oily grime and pull it off hard surfaces.
Heat, changes in pH, and even hard water don’t shake N,N-Dimethyloctyldecanamide too much. Standard sodium-based detergents can stop working well at high temperatures or in water packed with calcium, but this amide keeps working. Factories, food processors, and anyone trying to keep big kitchens spotless end up relying on surfactants like this because fewer variables translate to steadier cleaning results and less wasted product. That means fewer harsh chemicals get dumped down the drain because one ingredient stays effective longer.
Chemical companies pick this amide as a co-surfactant for a reason. It plays well with both ionic and nonionic surfactants, boosting overall stability and power. It won't foam up uncontrollably, which matters if you're running industrial machines that can choke on too many bubbles. As for toxicity, safety data shows moderate irritation if you soak your hands in it, but hazard levels fall well below stricter European chemical guidelines if handled with care. Every cleaning product should still include clear directions and hazard warnings—cutting corners doesn't just risk employee health, it can lead to regulatory fines.
It’s easy to overlook what happens after the job’s done and wastewater heads for treatment plants. Biodegradability sets this amide apart from many older cleaning chemicals. Studies and regulatory filings show that microbes in water treatment break it down instead of letting it build up. Companies using this compound can keep pace with growing demand for greener formulations, which satisfies both end users and government rules tightening up every year. Distributors who have switched over tell me the process is pretty seamless—just some tweaks to ratios and application methods.
Demand keeps growing for cleaning agents that do their job without sticking around in soil or rivers. N,N-Dimethyloctyldecanamide doesn’t solve every problem, but its properties keep it ahead of many petrochemical leftovers. Manufacturers can further reduce hazard by bundling it with plant-derived surfactants and using less overall product per cleaning cycle. Improved labeling and education around safe handling will help both employers and workers adapt, especially as chemical regulations keep evolving.
In my experience, a smart approach blends good science, practical packaging, and real-world training. With all the pressure to hit sustainability targets and keep environments sterile, this amide offers one tool that doesn’t call for trade-offs between performance and environmental health.In the back rooms and labs across industries, N,N-Dimethyloctyldecanamide keeps showing up in surfactants, cleaners, and specialty process aids. It’s one of those compounds that nobody outside the field talks about, yet it stays in heavy rotation once you know what it does. With chemicals like this, storage might not spark headlines, but small mistakes can get expensive—fast.
Experience in warehouses, not just reading labels, teaches a few clear lessons. N,N-Dimethyloctyldecanamide prefers a cool, dry spot. Moisture, heat, or light can mess with its composition. Some old timers would shove all surfactant compounds on the same shelf, thinking “these don’t explode—so what’s the real risk?” Turns out that shortcut can lead to sticky residue, odor changes, instability, or even a ruined batch. Specialty companies warn about cross-contamination for good reason. Keep it capped, keep air out, and keep the drum or container off the floor. Once water sneaks in, the quality slides.
Always reach for high-density plastic drums or steel containers. Both block out most ambient moisture and take a beating in transit. In a tight space, it’s tempting to stack barrels two or three high. Just remember, tipping equals loss—spilled N,N-Dimethyloctyldecanamide can cause slip hazards and won’t clean up easily. Label everything in plain language and make sure date stamps show when that batch hit your supply.
Leaving chemicals in a hot or unventilated room? Short path to volatility. Long-term, heat triggers slow breakdown. A surprising number of storage rooms tip over 30°C in the summer, especially in older buildings. Find a shaded spot, with ventilation, and keep temperature steady. Fluctuations don’t help either. In my own shop, we discovered a few years back that one shelf under a skylight created inconsistent results, spoiling additive blends we needed for a contract just because sunbeams hit it every morning.
If condensation creeps in—maybe from poor sealing or a random uptick in humidity—you end up with emulsification, off-odors, or product separating out like tired salad dressing. Dehumidifiers are a smart investment in any chemical workroom. Removing that variable cuts troubleshooting later.
People come first. Safety data sheets call for gloves, goggles, and keeping skin contact to a minimum. In real life, it’s easy to get sloppy after the first dozen transfers or decants. Training helps, but so do clear signs and having gloves in arm’s reach. Keep spill kits stocked and make sure everyone on your team knows how to use them. Never pour leftovers down the drain—this stuff builds up in water systems. Lock up the storage area away from food, drink, and break zones.
I’ve seen plenty of businesses treat chemical storage as an afterthought, then get stuck with waste, lost revenue, or even health complaints. Smart storage for N,N-Dimethyloctyldecanamide really just means giving it the same respect as you give profit margins—go the extra step, keep things dry and contained, keep your people informed. In the long run, the cost of doing it right comes in way under the price of avoidable mistakes.
N,N-Dimethyloctyldecanamide gets added to many cleaning products and industrial fluids for its ability to break down oils and grease. Its role in helping clean up oily messes sometimes brings up questions on what happens to it after the scrubbing is done. As someone who has spent years studying surfactants and their impact in both industrial labs and local waterways, I’ve seen how even a small amount of a persistent chemical can change the environment downstream.
Claims about biodegradability often show up on product labels, but real-world breakdown depends on more than just chemistry textbooks. N,N-Dimethyloctyldecanamide shows some promise here: laboratory studies suggest bacteria can degrade it under certain conditions. Some research points to decent breakdown over a few weeks when conditions encourage microbial activity, meaning oxygen, warmth, and an active population of bacteria. But that’s not always the case outside the lab.
Out in a city or rural environment, wastewater plants do the bulk of breaking down chemicals before water returns to rivers. The issue? Many treatment plants measure efficiency using well-known pollutants, not newer cleaning agents. If N,N-Dimethyloctyldecanamide slips past, it winds up in streams and soils, where breakdown might slow to a crawl. This creates a situation where a product sold as “biodegradable” in one context lingers for months or even years in another.
Getting into rivers and lakes, anything slow to decompose can pile up over time. This is more than a theoretical risk. I remember walking a small Ohio creek in midsummer, only to spot unnatural foam in places where municipal outflow hit warmer currents. Surfactants, whether natural or synthetic, can change the balance for the fish and tiny microbe life that keep these ecosystems in check.
N,N-Dimethyloctyldecanamide doesn’t pack the same punch as the notorious “forever chemicals,” but that shouldn’t lower the standards for scrutiny. Some studies suggest it has low toxicity for fish and aquatic invertebrates, at least at typical exposure levels. Still, long-term buildup from daily discharges matters — especially where households, hospitals, or factories commonly flush water treated with similar ingredients. Without ongoing monitoring, small concentrations today may spell disruptions a year or two down the line.
Companies marketing cleaning products often lean on biodegradability as a positive selling point. Yet the term lacks a standard definition across the globe. Something can pass a regulator’s test for breaking down within a standard time, but that same substance may degrade slower in a chilly northern stream than in a carefully controlled test tank. Local climate, diversity of bacteria, and chemical load from other sources all make a difference.
Lower-impact cleaning ingredients exist. Certifications for safer chemistry, like EPA’s Safer Choice, set a higher bar for testing environmental fate and toxicity. Pushing manufacturers to submit chemicals for such independent review encourages honest labeling. Municipal water treatment and green chemistry researchers should keep a close eye on how new substances like N,N-Dimethyloctyldecanamide behave beyond the lab, especially as formulas and environmental regulations evolve.
Making safer products means listening to both scientific evidence and community feedback. Every part of the chain — from chemical producers to the person pouring detergent down the drain — has a role to play in reducing lingering environmental risks.