Tetrabutylammonium nitrate did not show up by accident in chemistry labs. Its early days trace back to the growth of quaternary ammonium salts, primarily used for their role as phase-transfer catalysts. In the mid-1900s, researchers began to focus harder on organic nitrates for their unique reactivity, looking for ways to make reactions smoother and faster, particularly those that demanded non-aqueous conditions. Tetrabutylammonium nitrate became a subject of interest as scientists started addressing the need for soluble nitrate sources in organic media. Along the way, discovery was driven by practicality—chemist sought reagents that would cleanly deliver nitrate ions where classical inorganic salts fell short, mainly due to their poor solubility in organic solvents. By the 1970s, its preparation methods were documented in journals, paving the way for it to find its niche in both research and industrial settings.
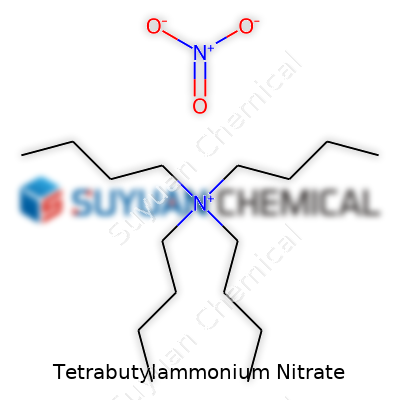
This compound looks simple on the surface—a combination of a big, alkyl-heavy cation and the nitrate anion. Yet that pairing creates a salt that dissolves easily in organic solvents and acts as a mild, reliable source of nitrate ions. The tetrabutylammonium framework stabilizes the nitrate and makes it easier to handle compared to more reactive or less soluble counterparts.
Physically, tetrabutylammonium nitrate presents as a white, often crystalline solid if properly purified. It holds up well under room temperature, though it can slowly absorb water from moist air because of its hygroscopic tendencies. It carries a relatively high molecular weight due to the four butyl chains hanging from the nitrogen. In terms of chemical traits, the compound demonstrates strong ionic character, which translates into good solubility in a wide array of organic solvents—dichloromethane, acetonitrile, even alcohols. Thermal stability stays decent up to moderate temperatures, but things fall apart quickly around 200°C, with risk of nitrate decomposition and potential release of nitrogen oxides.
Industry usually calls for a product with purity north of 98%. Presence of moisture, heavy metals, or organic contaminants can undercut its reliability, especially in sensitive reactions. Labels should give lot numbers, purity, storage instructions, and hazard warnings. Because the nitrate group carries risk, both chemically, and in some countries, legally, documentation needs to be tight—clear hazard pictograms, risk phrases, and guidance for handling and first aid.
Laboratory synthesis begins with tetrabutylammonium bromide or chloride. The salt reacts with silver nitrate in water or alcohol. Silver halide falls out, while tetrabutylammonium nitrate stays dissolved. Filtration removes the precipitated silver salt, and a round of solvent evaporation or crystallization follows to pull out the nearly pure nitrate salt. This route works well for bench-scale preparation, but silver nitrate costs scale poorly for production. Alternative methods involve ion exchange resins, where a loaded resin swaps out the halide for nitrate in a batch process. Some plants may go with direct metathesis methods, using sodium nitrate and tetrabutylammonium halides, followed by careful separation and drying.
Once in hand, tetrabutylammonium nitrate can step up in many transformations. In organic synthesis, it serves as a nitrate donor, supporting nitration reactions that otherwise fail in low-polarity media. It mediates the oxidation of alcohols, converts alkenes, and even works in biological contexts, particularly for release of nitric oxide under certain conditions. As a supporting electrolyte, it allows careful study of redox transformations, giving inert ions that do not interfere with the chemistry at the electrode. Researchers also tinker with the cation—some trade butyl for methyl or ethyl chains to alter solubility or melting points, applying similar thinking across the family of quaternary ammonium nitrates.
Tetrabutylammonium nitrate shows up in catalogues and studies under several names. Some use "TBA nitrate" or abbreviate as "TBANO3." Others list the longer variation: "Nitrate, tetrabutylammonium salt." Researchers sometimes shorten it to "NBu4NO3," keeping things tidy in crowded tables or reaction schemes. No matter the label, the structure remains constant—four butyl arms on a nitrogen, balancing out a single nitrate.
Handling needs respect. The nitrate anion brings its own risks, especially under heating or in combination with combustible materials, since it can act as a mild oxidizer. Companies spell out personal protective gear—lab coat, nitrile gloves, splash goggles. Access to a functional fume hood is a must, as is a set of dry chemical extinguishers nearby. Workers should keep the salt stored cool and dry, away from acids, bases, and reducing agents. Factory protocols demand spill kits, emergency eyewash stations, and detailed incident logs. Waste management falls under local hazardous materials regulation, typically handled by licensed disposal contractors. Its potential use in illicit activities brings security concerns, too, with certain jurisdictions flagging sales for close monitoring or purchase limits.
Research labs value tetrabutylammonium nitrate for its role in organic synthesis. It’s a favorite pick when traditional inorganic nitrate salts just won’t dissolve. Its presence in phase-transfer catalysis brings particular benefit to multi-step syntheses, including heterocycle construction and green oxidations. It comes to the rescue as an electrolyte in non-aqueous electrochemistry, letting researchers probe electrode reactions without interference from sodium or potassium ions. In emerging areas like ionic liquid design, the basic tetrabutylammonium architecture inspires new salts with customized properties for battery and sensor research. Some corners of pharmaceutical chemistry explore it as a mild nitrating agent to introduce nitro groups without harsh methods. Teaching labs lean on it for demonstrations, letting undergraduates get firsthand experience in phase-transfer and ionic reactivity.
Chemists keep searching for new roles for tetrabutylammonium nitrate. Interest has grown in green chemistry, focusing on reaction media that avoid classical mineral acids and provide safer, easier pathways to complex molecules. Industrial research explores its function in hybrid catalysis, using it to shuttle ions between incompatible phases. Environmental researchers evaluate its behavior in soil and water systems, tracing its breakdown and movement in both controlled and real-world scenarios. Some startups have tested its fit in electrolytes for high-performance supercapacitors, eyeing stability and compatibility with carbon electrodes. At conferences, one can usually spot a poster testing a new use for this compound, whether in renewable energy, advanced separations, or microfabrication.
Toxicity does not get overlooked. Most data so far suggests that tetrabutylammonium nitrate brings moderate risk in mammals, mainly due to its nitrate component. High doses have shown to induce methemoglobinemia, harming the ability of blood to carry oxygen. No evidence ties it directly to genetic harm or cancer, but long-term exposure limits remain under-studied. Environmental impact tests show a need for caution—if released in bulk into water or soils, the nitrate can fuel algae blooms or disrupt aquatic life. Safety protocols build in redundancy—engineers track air quality, monitor dermal exposure, and require medical monitoring for long-term users. Detailed data sheets go through layers of regulatory review before products reach market.
Demand for tetrabutylammonium nitrate looks set to grow, especially as advanced materials and sustainable chemistry take center stage. Scientists want reagents that deliver selective reactivity without harsh byproducts, and this salt fits the bill for many transformations. Companies seek to develop more sustainable production routes, ideally cutting reliance on limited or expensive resources like silver nitrate. Regulatory pressure for greener processes should keep research focused on lowering waste and improving biodegradability. As more industries embrace ionic liquids, alternative batteries, and eco-friendly oxidations, the tetrabutylammonium backbone will likely serve as both a template and a tool for designing safer, better, and more effective technologies. Meanwhile, responsible stewardship—safe storage, documented use, and careful disposal—will remain essential as this niche compound becomes a bigger presence across laboratories and production sites worldwide.
Tetrabutylammonium nitrate, or TBAN for short, isn’t one of those compounds you spot on a list of household products. This chemical pops up in labs, research institutions, and sometimes in industries looking for very specific results. Its structure combines a nitrated ion with a hefty organic cation, giving it properties that chemists count on, especially when they want to swap ions around or stir up reactions without water causing trouble.
In my years of working alongside research chemists, I noticed TBAN coming off the shelf during tricky syntheses—particularly in organic reactions where water ruins progress. Its salt dissolves easily in many organic solvents. This ease-of-use gives teams more control than old-school inorganic salts, which often gunk up or don’t dissolve. TBAN helps deliver nitrate ions where needed, supporting processes ranging from oxidations to tricky substitutions.
A good example of TBAN’s value appears in the fine details of making new molecules for experimental drugs. Sometimes, a research team tries to add a nitrate group for testing a new medical compound's efficiency or selectivity. Purity and control matter here. TBAN avoids introducing unwanted metals or impurities. Teams know what’s in the mix, which matters when results have implications for human health.
Electrochemists use TBAN salts as electrolytes because their stability holds up against high voltages and temperature shifts. I’ve seen researchers turn to TBAN for testing prototype batteries. The salt doesn’t degrade or form weird byproducts. This difference lets energy storage engineers study reactions that mimic future real-world batteries, and get reliable numbers when efficiency can’t slide by even one percent.
Besides energy research, TBAN pops up during chemical analysis in the lab. It’s a favorite for liquid–liquid extraction and for salting out reactions that need a nudge to form clean, separated layers. In my own work purifying tricky samples, swapping in TBAN sometimes led to much faster separation and less mess to clean up—a relief when chasing tight research deadlines.
TBAN isn’t without headaches. Safety data sheets show that a careless spill or inhalation can bring on health problems. It’s important to keep solid procedures, ventilate the workspace and wear proper protection. Supply can fluctuate or be pricey since it's not produced on the same scale as more common salts.
Environmental impact deserves attention too. Like most organic ionic compounds, disposal matters. Allowing these chemicals to wash away untreated adds up, especially as more labs use them.
Researchers keep finding more precise uses for TBAN. Organic chemists want greener, safer solvents and reagents. Battery engineers crave reliable testing environments. Medical chemistry leans hard on ultra-pure inputs. Tetrabutylammonium nitrate steps up in these labs, offering flexibility, purity, and predictability. As regulations tighten and costs matter more, teams keep pushing for smarter processes and safer handling—turning niche chemicals into workhorses for tomorrow’s science.
Tetrabutylammonium nitrate carries a name that’s a mouthful, but its story goes much deeper than a string of scientific jargon. At its root, you find the formula C16H36NNO3. Each part of that code matters, from the butyl chains to the nitrate partner. I’ve found that studying chemicals like this unlocks a whole world of practical chemistry, and sometimes, knowing what those letters stand for sheds light on the way chemistry shapes the way we build, clean, and invent.
The formula C16H36NNO3 spells out the basics: sixteen carbons, thirty-six hydrogens, two nitrogens, and three oxygens. Four straight butyl groups surround a central nitrogen, pairing up with a nitrate anion. This structure means Tetrabutylammonium nitrate behaves differently than that salt in your kitchen. In laboratories, I’ve seen it act as a phase transfer catalyst, moving ions between water and organic solvents. That’s not something ordinary table salt can do.
Lots of students remember formulas by rote, but I learned how much easier solving chemical puzzles gets when you can picture what the atoms are doing. That’s especially true for quaternary ammonium salts like this one. In labs and classrooms, explaining this formula isn’t just about hitting the exam mark—it’s about understanding why this compound dissolves in organic solvents, why it carries ions where others can’t, and why its hydrophobic tail and ionic head make a recipe for chemical transformation.
Outside textbooks, people use Tetrabutylammonium nitrate for organic synthesis, pollution cleanup, and sometimes in explosives research. Because it carries both a bulky, organic cation and a reactive nitrate, you’ll find it bridging the gap between oil and water in the lab. So many reactions fail if you can’t get the necessary parts together in the same spot; this compound solves that problem in one fell swoop.
With its value comes a level of responsibility. Any chemical that brings together big, easy-to-manipulate cations and energetic anions requires careful handling. Nitrate’s reactivity opens doors, but it can also cause headaches if storage gets sloppy or disposal is careless. I’ve always paid close attention to safety protocols—especially with chemicals like this—and every chemist working with Tetrabutylammonium nitrate should do the same. The more you know about its formula, the more you respect that balance between usefulness and risk.
By looking at a formula like C16H36NNO3, there is a lesson: chemistry isn’t warehouse math; it’s active understanding. High school classes rarely dive into chemicals with names this long, but once you do, the entire landscape of possibilities changes. Advocating for clearer chemical education stands out as one answer—one that equips students and professionals alike not just to repeat formulas, but to make sense of them in practical, safe, and creative ways.
In any lab, Tetrabutylammonium Nitrate brings a mix of opportunity and risk. Its uses pop up in organic syntheses and phase-transfer catalysis, but the nitrate part makes the chemical much more than just another reagent. Most folks working in a lab remember the stories about nitrates, and for good reason—similar substances have fueled accidents due to poor judgment or relaxed habits.
Lots of major incidents start with ignoring the heat. Flammable materials and oxidizers do not forgive carelessness. Tetrabutylammonium Nitrate does break down and lose stability when temperatures climb. I remember a colleague mentioning a container stored too close to the top of a shelf, just above a heat vent. After a few months, the outer container started to show signs of stress—slight swelling and powder caking at the seal.
The answer? Cold, dark storage works best. Use a dry cabinet in the temperature range between two and eight degrees Celsius, far from direct sunlight, radiators, or compressors. The cooler, the better, assuming the substance won’t freeze solid. Keeping it away from heat sources feels like common sense, but so does checking for any loose seals or cracks. Many labs rotate containers, with oldest to the front. It may seem tedious, but enough forgotten bottles have caused problems to make rotation a worthwhile routine.
Moisture turns a small risk into a big one. Tetrabutylammonium Nitrate attracts water from the air due to its hygroscopic nature, and every extra drop increases decomposition rate. Never trust a screw cap alone. Go for double containment: tightly closed glass bottles, sealed inside plastic bags with desiccant packs thrown in for good measure. I’ve known folks who wrap tape around vial necks and lids—old school, but effective.
Too many spills come from the assumption that a bottle cap can handle years of humidity swings. Routine label checks and examining tightness of closures stop disasters before they start. In places with regular lab audits, this task becomes second nature. When no one checks, the risk goes up with each month that passes.
Storage location means everything. Tetrabutylammonium Nitrate counts as a strong oxidizer. It never belongs with organics, reducing agents, acids, or anything flammable. In the labs I’ve worked in, we separated oxidizers onto special metal shelves lined with corrosion-resistant trays, far from solvent cabinets.
Color coding goes a long way. A brightly marked shelf or well-labeled tray keeps anyone from mixing up bottles at a glance. Training new folks to double-check those color codes saves headaches and prevents cross-contamination. For busy shared labs, marked secondary containers prevent worst-case scenarios.
Regular inventory checks and staff reminders usually create fewer emergencies. Training every new lab member on storage rules, rather than just a single orientation before they ever see a bottle, helps avoid “quick fix” shortcuts. A document by the storage area listing the basic rules—temperature, dryness, no sunlight, no open flames—shapes habits over time.
Relying on a checklist rather than memory gives everyone a better chance of catching mistakes before chemicals spoil or react. Combining practical training, routine inspections, and clear physical boundaries keeps tetrabutylammonium nitrate—and everyone handling it—much safer.
Tetrabutylammonium nitrate shows up in research labs, in organic synthesis work, and sometimes as a phase transfer catalyst. It’s a white, crystalline salt that usually looks plain, but can catch many off guard who aren’t used to handling powerful reagents. You won’t find it in everyday consumer products, but chemists and lab workers use it for making certain chemical reactions run smoother.
This salt hits a tricky intersection: its nitrate group brings oxidizing strength, enough to make it potentially dangerous. Nitrate salts have a record for reacting strongly—sometimes explosively—if mixed with organic stuff that loves to burn. News trickles through academic channels about near misses and loud pops when someone treats these compounds like table salt instead of a chemical hazard. No one wants a runaway reaction in a crowded laboratory.
Breathing in dust, getting it on your skin, or getting it in your eyes often means irritation. Repeated, careless handling ramps up risks. Some studies flag butyl ammonium compounds for acute toxicity in rodents, turning up symptoms after exposure—trouble breathing, damage to organs, sometimes affecting the nervous system. Even if big, long-term studies in people don’t line up, the signs in the lab are enough to keep the gloves on and the windows open when working with any concentrated ammonium salts, nitrates included.
Tetrabutylammonium ion breaks down slowly. Dumping it down the drain or into the soil means it can build up in the environment, possibly messing with aquatic systems and soil microbes. Aquatic life, especially small invertebrates, show sensitivity to quaternary ammonium salts. High concentrations can promote fish kills, disrupt plankton communities, and tip the balance in waterways. It doesn’t belong in the ecosystem, and most research facilities train people to keep it contained and disposed of through chemical waste streams.
I’ve watched folks in research settings cut corners and get lucky—not the kind of luck anyone wants long-term. Clear rules help: wear gloves and eye protection, work with fume hoods, and never eat or drink anywhere near lab benches. Chemical hygiene plans spell these things out, but real world stories drive the lesson home far more effectively than binders full of protocols.
Fire risk usually feels remote, until it isn’t. Using glassware free from cracks, keeping flammable solvents away from nitrate powders, and storing the salt in a cool, dry spot far from acids or reducing agents helps put distance between safe practice and stories told by accident investigators.
Every hazardous material comes with a learning curve. For Tetrabutylammonium nitrate, sticking to proper waste disposal routes protects both workers and the local water supply. Running short training sessions and sharing stories on lab safety at regular intervals keeps people sharp. Using smaller batches and considering less hazardous substitutes keeps the risk lower in the first place.
Science rewards curiosity and caution in equal measure. Tetrabutylammonium nitrate doesn’t cause problems when treated with the respect any strong oxidizer deserves, good ventilation, careful storage, protective equipment, and proper disposal. The end goal is always the same—keep people healthy, protect the world outside the lab, and get the chemistry done right.
Walk into any chemistry lab and you’ll notice just how often reagents quietly make or break an experiment. Tetrabutylammonium Nitrate, or TBAN for those who spend their days at the bench, comes up frequently for phase transfer catalysis, organic synthesis, and electrochemistry. The thing nobody likes admitting? Purity matters. More than most are willing to say. And the reality doesn’t always match the pristine promise slapped on the bottle label.
Most commercial TBAN sits around 97% to 99% purity. Some catalogues dangle “99%+” numbers, but experienced scientists will tell you: always double-check the fine print. Some brands cater to high-end research, others to bulk production. Everything depends on source, manufacturing process, and storage. Despite all claims, trace impurities crop up in nearly every batch. Expect water content, bits of unreacted tetrabutylammonium salts, and sometimes leftover organic solvents from processing.
The reality is plain: even tiny impurities can steer a project off course. If you’re working on something sensitive, like mechanistic studies, these stray molecules eventually show up in your results—sometimes as ghost peaks on an NMR, other times as flaky yields that randomly tank.
Take personal experience: back in grad school, a routine synthesis using off-the-shelf TBAN suddenly started giving inconsistent yields. Hours poured into debugging solvent, glassware, atmosphere—nothing explained it. Only after a run through ion chromatography did the truth come out. The “98% pure” TBAN contained nearly 0.7% chloride, a leftover from incomplete purification. The supplier shrugged when we called. “Meets the spec.” Sure—but nobody in the lab trusted that lot again.
Purity doesn’t just matter for the sake of neat paperwork. A trace halide and an unnoticed water content can completely redirect selectivity, reaction rates, or catalysis. Electrochemistry, for example, is notoriously unforgiving—an unknown cation or drop of water skews current efficiency and sometimes corrodes electrodes. Even in organic transformations, what seems like a mere few tenths of a percent can mean the difference between a clean isolation and endless purification steps.
Nobody should have to gamble on a reagent. The best chemists I know verify every batch. Sending out a sample for in-house analysis—anion testing, water analysis, and even a simple NMR scan—saves time and credibility down the line. Some universities go so far as to only use TBAN freshly recrystallized in-lab, drying it in vacuo to wring out any water left hiding after shipping. For work of particular sensitivity, in-house purification isn’t just encouraged, it’s written into protocols.
Suppliers do respond to pressure. Enough calls from frustrated labs force catalogues to publish more complete certificates of analysis. Open data from third-party testing adds transparency, which slowly pushes quality up across the board. If your institution doesn’t already demand batch analysis on high-purity salts and TBAN, it’s worth sending a memo.
Trust in research often comes down to the small stuff. TBAN’s purity marks only one example, but it illustrates why experienced chemists combine common sense with skepticism. In real-world chemistry, certificates are not enough. Independent checks, open reporting, and a feedback loop with suppliers provide a clear path forward. For anyone serious about reproducible work, treating even “high-purity” TBAN as an ingredient that deserves scrutiny never lets you down.