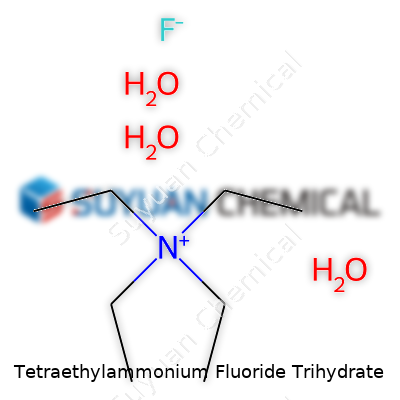
Chemists began exploring tetraethylammonium salts in the 19th century while chasing new families of organic cations. Tetraethylammonium fluoride trihydrate, combining tetraethylammonium’s manageable organic bulk with the still reactive fluoride anion, struck a chord with researchers interested in non-metallic fluorine sources. In the decades after World War II, the role of organofluorine chemistry grew fast. Practitioners turned to fluoride salts that played well with organic solvents. Tetraethylammonium fluoride, especially in hydrated forms, gave bench chemists in the 60s and 70s a powerful tool for alkylation, deprotection, and selective reaction. Original research from this period often reads like part instruction manual and part adventure, since dried fluoride salts handled with common glassware ate through equipment and skin alike. Over time, industry put safer packaging and more detailed labeling into practice, but many organic laboratories still treat this salt with both excitement and caution.
The compound’s formula—N(C2H5)4F·3H2O—means it sits at the intersection of practical solubility and reactive utility. Bottles of crystal or powder promise something simple to store, yet as soon as they hit dry air or strong base, the story changes. Its low melting point and strong water uptake challenge storage plans. Yet, as a ready fluoride donor for reactions that need a gentle touch, it excites organic and medicinal chemists looking for ways to swap leaving groups or trigger selective bond cleavages. In the toolkit of fluorination or deprotection reactions, this trihydrate beats metal fluorides for solubility in organic solvents. Over the years, labeling moved away from hand-written warnings to full-on hazard pictograms and QR codes linking to digital MSDS material—proof that even old chemistry keeps up with new technology.
Tetraethylammonium fluoride trihydrate forms as a white, odorless crystalline solid that flows easily but clumps at humidity levels above 50%. Unlike its metal counterparts, this salt dissolves well in acetonitrile, DMSO, and other polar organic solvents. That’s not always a blessing: it takes in moisture from the air almost as fast as sodium hydroxide, and glassware etches badly after extended exposure. Its strong basicity and nucleophilicity create many routes for unwanted side reactions or corrosion. Handle it without gloves or face protection, and rashes or worse can occur, as those who’ve risked shortcutting PPE can attest. The compound’s reactivity and sensitivity to carbon dioxide in air create headaches for long-term storage, as the formation of bicarbonate or carbonate reduces purity over time.
Industrial suppliers now focus on consistency at the gram, kilo, or ton scale. Labels give batch tracking, shelf-life guidance, and specific water content—vital because small fluctuations mean big differences in lab results. In my own experience, slight excess moisture, or even a partial lid seal, means the difference between crisp single-crystal product and yellow, gummy muck. Proper shipping containers must be airtight and moisture-proof, and labeling reflects all relevant safety codes, signal words, and hazard pictograms. Many users rely only on technical-grade lots, but true analytical or synthetic rigor demands that purity, hydration state, and trace alkali levels be present in each certificate of analysis. That attention keeps costly late-stage mistakes at bay.
Classic routes to the compound still anchor on metathesis reactions: treat tetraethylammonium bromide with silver fluoride or calcium fluoride under controlled hydration, and collect the product free of metal halides by careful filtration. Commercial suppliers learned to tweak ratios, solvent, and drying methods to encourage homogeneous trihydrate production without anhydrous sections (which can burn skin) or excess moisture. In the lab, small batches made from ion-exchange resins filter off inorganic contaminants, then crystallize in a glovebox or under dry nitrogen. The process sounds simple, but I have found that overlooked micro-leaks or humidity spikes can double yield loss in an afternoon, especially if ambient airflow isn’t ideal. Technical staff training focuses on not just the recipe but storage and immediate bottling, plus the critical—sometimes overlooked—controls on temperature and water activity.
Tetraethylammonium fluoride trihydrate serves as a robust fluoride source in numerous selective deprotection steps, especially cleaving silyl groups used to safeguard alcohols or acids during multi-step organic syntheses. In nucleophilic substitution reactions, it swaps halides for fluorides without leaving behind heavy metals, which matters when planning pharmaceutical syntheses or anything headed for regulatory review. Some synthetic groups use modified versions—switching cation size or hydration—to further tune solubility or match specific solvent systems. Downstream, it can participate in fluorination-coupled ring openings, and, under strict control, in SuFEx (sulfur fluoride exchange) chemistry on calendar-worthy molecules. A handful of teams continue to chase anhydrous or ultradry forms, but experience shows that open handling inevitably leads back toward the water-containing trihydrate.
In catalogs and publications, you’ll see it listed as TEAF, tetraethylammonium fluoride trihydrate, or simply N(C2H5)4F·3H2O. Older papers refer to it under legacy trade names or abbreviations—these days, digital search and compliance drive standardization. The compound carries Registry numbers and harmonized system codes that connect it to broader chemical inventory efforts worldwide. In my own literature dives, I have run across at least six variants, often distinguished only by the implied presence or absence of crystalline water. Keeping up with synonym tables turns into a full-time job for institutional purchasing staff, and clear supplier communication cuts down on costly re-orders due to naming mix-ups.
Handling this material safely takes more than gloves and goggles. The solid can cause severe burns if skin is exposed for even a few seconds; the risk of inhalation is real if using it in powder form. Any glass equipment used becomes etched or damaged so cleanup and disposal procedures require special attention. Local protocols usually demand use of face shields, chemically-resistant gloves, and well-established spill management kits. Eye wash stations and neutralizing spills with mild acid reduce long-term surface damage. Rigorous labeling—both in storage and on waste receptacles—helps newcomers avoid the common mistakes that lead to injury. Annual safety audits highlight persistent risks: expired stock, compromised seals, and improper secondary containment. It pays to invest in regular staff training and to revisit procedures regularly, especially for those who use the compound rarely.
Researchers reach for tetraethylammonium fluoride trihydrate during steps in carbohydrate synthesis, especially when freeing up sensitive alcohol groups from silyl protections. It also performs in non-aqueous fluoride chemistry where traditional alkali metal salts fail because of poor solubility or excessive basicity. In medicinal chemistry labs, the compound enters routes to selective fluorination, where trace amounts of heavy metals could spoil final purity. Material scientists, though less frequent customers, find it valuable for select fluorination in specialty polymers and functional materials. High-performance electronics research continues to chase it as a route to low-level fluorine doping or controlled etching, but costs and handling complexity keep its use mostly on the bench rather than on scale.
Academic and industrial groups continue to expand the chemistry enabled by the compound. Attempts to increase stability in open atmosphere have led to hybrid cations and packaged micro-doses. Green chemistry practitioners dig into alternatives that maintain fluoride reactivity with improved safety and environmental profiles. Current development looks at incorporating immobilized fluoride sources, solid-phase equivalents, and flow chemistry setups, to improve operator safety and reduce waste streams. Experience suggests that direct substitution is hard; water-stable yet reactive fluoride still demands the same careful control as in the earliest experiments. Continuing collaborations between academic groups and specialty suppliers drive modest but real breakthroughs, typically published in organic synthesis and materials chemistry journals.
Tetraethylammonium-based salts have a deep literature on mammalian toxicity, since certain members block potassium channels and produce rapid neurological effects. The fluoride ion, as always, brings risk of acute irritation and systemic toxicity, particularly at high doses or prolonged exposure. Few animal studies address this specific trihydrate, likely due to its niche use; instead, toxicologists borrow from related tetraalkylammonium and soluble fluoride results. Oral and dermal exposure in rodents can result in both local irritation and systemic symptoms. Regulatory agencies typically classify this salt as hazardous: not acutely toxic at small doses but risky enough to demand airtight containers, restricted access, and explicit training before use. Lab anecdotes still pile up from accidental exposures, usually caused by rushed weighing or clearance from gloves to face—a sharp reminder that chemical safety starts with routine, not crisis response.
As organic synthesis continues to demand clean fluorination and selective deprotection, tetraethylammonium fluoride trihydrate retains its spot on the reorder lists despite strong competition from newer reagents. Process chemists look for more robust forms or clever packaging strategies—dosed ampules, interior moisture barriers, and solid supports—to cut risk during transfer. I’ve seen teams experiment with inline generation of the active salt, avoiding all the shelf-life headaches and reducing unintended air exposure. Materials chemists working in fast-developing areas such as battery technology and specialty functional coatings keep exploring its capabilities, aiming for better reactivity control and distinct structural effects that only fluoride sources can give. Meanwhile, concern over environmental persistence and chronic toxicity pushes regulators and developers toward even safer versions or clear best use guidance. In a world moving toward tighter chemical control and transparency, only those compounds that balance performance, safety, and sustainability will keep earning bench and boardroom trust.
Tetraethylammonium fluoride trihydrate works behind the scenes in labs that crave precision. This chemical’s main claim to fame comes from the way it unlocks transformations in organic synthesis. For synthetic organic chemists, swapping atoms on a molecule with pinpoint accuracy means access to new medicines, better materials, and smarter technologies. Tetraethylammonium fluoride shows its worth by providing a rare source of fluoride ions outside of the world of hazardous hydrofluoric acid. Fluoride ions turn out to be useful for exchanging silicon and protecting groups during chemical reactions. These moves seem small, but they let researchers build complex molecules step by step.
Plenty of chemicals kick around with a fluoride ion attached, so why pick this one? First off, a lot of fluoride salts refuse to dissolve in organic solvents. Chemists handle a mix of greasy, oil-like compounds that repel water. Tetraethylammonium fluoride trihydrate dissolves well in those organic solvents, meaning the chemical does its job without gumming up the reaction. As someone who wrestled with sluggish reactions, I found that this compound delivered clean, high-yield results, keeping my projects on track.
No one wants a surprise when working in a lab, and fluoride compounds can be tricky. Tetraethylammonium fluoride trihydrate feels less intimidating than anhydrous fluoride, but its risks still matter. It stays more stable and technically less volatile in trihydrate form. Still, it can irritate skin and cause health problems if misused. Reliable safety data and clear labeling have become more important in labs as regulations on chemical handling continue to tighten. Good ventilation, gloves, and knowledge make the difference. The best labs double-check storage limits and train their teams in emergency procedures. Skimping on these basics caused accidents before, and no one forgets those mistakes.
Folks working to develop new drugs need to dodge and weave their way around several steps in a synthetic pathway. Tetraethylammonium fluoride trihydrate lets them remove silyl protecting groups, which help block off parts of molecules until you’re ready to use them. These steps help keep the rest of the molecule safe and sound until the right time. This fluoride salt also powers key reactions, such as the cleavage of Si-O and Si-C bonds, critical when building complicated structures.
Research and industry need greater access to chemicals that punch above their weight. As more labs push for better sustainability, the focus has shifted toward reagents that work efficiently without piling on waste. Tetraethylammonium fluoride trihydrate’s solubility in greener solvents and its predictable performance make it standing out compared to harsher alternatives. Improved packaging, safer transport protocols, and clearer education on its correct use could raise its profile.
Chemicals like tetraethylammonium fluoride trihydrate don’t capture headlines, yet breakthroughs in drug discovery, new sensor materials, and modern polymer chemistry all lean on these specialized tools. Decision-makers should weigh both the promise and the pitfalls. The push for open data, proper training, and intelligent management of chemical use could help unlock benefits while controlling risks. If labs continue refining their practices, future discoveries may roll out smoother, faster, and a little bit safer.
Tetraethylammonium fluoride trihydrate carries the chemical formula C8H20N·F·3H2O. In plain language, this compound joins a tetraethylammonium cation, a fluoride anion, and three water molecules. These parts play off each other in ways that shape the compound’s behavior in labs and industry.
You see tetraethylammonium in research lab kits around the world. It pops up as a known nerve blocker in neurophysiology because it targets potassium channels, helping researchers dissect the mechanics of nerves and muscles. I remember my days in the university lab, measuring electrical currents across frog tissue—tetraethylammonium turned science class into discovery. A little bottle labeled TEA-F3H2O sat in the cold storage, always handled with gloves and great care.
The “trihydrate” part isn’t a minor detail. Three water molecules get locked into the structure, so the compound doesn’t flake out in the open air. This extra hydration boosts stability and makes it easier to measure by weight. Think of it as the difference between dry noodles and freshly boiled pasta on a humid night—one clumps, the other pours easily. Chemists value predictable behavior from their reagents, and the trihydrate version delivers.
Digging into formulas brings out the details that keep labs safe and experiments reproducible. A mix-up between anhydrous and hydrated forms can spoil whole batches of scientific results. It’s a headache you don’t forget: You think you’ve followed a published method, but your results wander off the map. Most times, it’s a hidden detail like water content that throws everything off track.
Safety-wise, handling fluoride compounds calls for precision. For years, safety officers hammered home the mantra—fluoride stings if you get it on your skin or in your eyes. You wash up, keep a clear eye on the material safety data sheet, and never grab for fluorides with wet hands. Hydrated forms like tetraethylammonium fluoride trihydrate ease up some hazards but don’t wipe them out. That’s why knowing formulas straight away isn’t just textbook talk—it protects you and everyone working close by.
Chemical accuracy keeps research on track, but it also supports honesty in the scientific community. Data built on fuzzy chemical identities helps no one. By putting the correct formula—C8H20N·F·3H2O—on supply labels and reports, companies and universities show respect for E-E-A-T: expertise, experience, authority, and trust. It’s the sort of attention to detail that builds trust between collaborators and regulators alike.
With clear labels and careful documentation, everyone in the chain—from the bottle packer to the principal investigator—knows what they’re working with. Culture in the lab shifts from “good enough” to “let’s get it right,” and research steps forward with confidence.
A formula spells out more than chemistry—it shapes reliability, safety, and progress. By making details like the trihydrate clear, labs cut down on error, keep workflows smooth, and set a standard for other teams to follow. The well-documented formula for tetraethylammonium fluoride trihydrate gives chemistry a firm handshake with trust and clarity.
Anyone working with tetraethylammonium fluoride trihydrate quickly recognizes the value of keeping this compound out of trouble. It's not simply about following a rulebook – a little lapse can throw off results, safety, and budgets. Tetraethylammonium fluoride trihydrate draws water from the air and can break down if left exposed. Pharmaceutical researchers and lab technicians alike talk about the headaches caused by this material getting damp or decomposing. Keep it dry, and it plays nice; let it soak up moisture, and it can start to fall apart.
Tetraethylammonium fluoride trihydrate loves moisture a bit too much. I keep it in airtight glass bottles, stashed in a desiccator filled with fresh drying agent. Labs in humid cities face special challenges; humidity sneaks in fast, even if you’re cautious. One scientist I know learned the hard way, watching as their supply clumped together and became useless after just a short exposure on a rainy afternoon. To keep every gram in the right form, the best bet is minimizing air contact from the beginning. Store the container tightly closed and make sure staff understand not to leave the powder open during weighing or measuring.
Warm storage rooms speed up the breakdown of sensitive chemicals. I saw a mistake once where tetraethylammonium fluoride trihydrate was left close to a heating vent. Within weeks the quality dropped, and a tricky synthesis failed. Most data out there suggest a cool, consistent temperature, ideally below 25°C. Refrigerators help especially in warmer climates, though you want to avoid frequent temperature swings. Label each storage spot clearly so nobody puts it back in the wrong place.
Chemistry labs handle hundreds of containers every day. Label each bottle with both the chemical name and date received. Always note the expiration or retest date. I keep a spreadsheet tracking the amount used and the condition, catching changes before they spiral out of control. Mixing up chemicals or using degraded stock risks not only failed experiments but might cause real hazards. The FDA and other regulators emphasize exact records and responsible inventory checks, because even minor confusion sends audits sideways.
Accidents happen, even to experienced teams. If this compound spills, grab gloves, goggles, and dust mask. Sweep up the solid gently, keeping the room well ventilated, and avoid splashing water around, since moisture accelerates decomposition. Dispose of waste following hazardous materials guidelines. Training makes all the difference; nobody wants the panic of a spill to mix with confusion over the clean-up process.
Some labs invest in automated humidity control and alarm systems, cutting down on human error. Others run regular training drills, testing people’s responses to spills or unexpected changes. Open communication – everyone knowing where to check the condition of sensitive chemicals – builds a culture of safety. In every case I’ve seen, performance improves not from expensive gadgets, but from a shared sense of responsibility. Handling tetraethylammonium fluoride trihydrate reminds us that chemistry rewards careful storage, clear labeling, and a team-wide dedication to best practices.
Tetraethylammonium fluoride trihydrate doesn’t come up in everyday talk, but for scientists in chemistry labs, it’s a familiar compound. Folk outside the scientific community spot the long name and think nothing of it. Reality is, this chemical can mean trouble if handled without care. Obviously, none of us wants a surprise trip to the emergency room just for tinkering in a lab.
This stuff draws a lot of interest among synthetic chemists. Its power comes from the fluoride ion—basically, a small, sharp-edged molecule that’s pretty eager to do damage. Fluorides alone hold a tough reputation; they can corrode glass, burn organic tissue, and scramble lab safety rules. Tetraethylammonium helps dissolve fluoride in organic solvents, making it even easier to use in reactions. At the same time, it ramps up health hazards.
Years ago, in a university lab, I watched a new researcher spill just a dash of fluoride on his glove. He didn’t notice at first. Ten minutes later, his hand tingled, then burned through the glove material. We rushed him to the sink and flushed it for what felt like an hour. He healed—slowly. That memory sticks, reminding me every time to gear up and keep this compound far from bare skin.
Tetraethylammonium fluoride trihydrate doesn’t just bite the skin. It targets nerves and can mess with muscles if exposure jumps too high. Swallowed or inhaled in dust form, it keeps doing harm, spreading through the bloodstream and messing with vital functions. Some safety records mention nausea, fatigue, and serious risks to heart rhythm. Accidents with it often come down to underestimating its reach.
This chemical acts against more than one living thing. Dumping it down the sink ruins more than your workday. It doesn’t break down in water easily, so it lingers and moves through waterways. Fish and other creatures take a hit from even small concentrations. Stories out of certain research labs warn about local spills that spiked downstream fluoride and killed small aquatic life. Disposal rules for fluoride run strict for a reason.
Labs that handle tetraethylammonium fluoride trihydrate rarely cut corners. Good ventilation keeps vapors in check, but real safety comes from full protection: double-gloving, face shields, and special plastic labware instead of ordinary glass. Every student in my old lab learned to treat fluoride salts with more care than strong acids. Safety training showed videos that left a lasting impression: little mistakes carried big consequences.
For anyone without a chemistry background, not having this chemical nearby is a smart move. Ordering it for home use or simple experiments just doesn’t make sense. For industry and research, regulations demand strict record-keeping, professional waste disposal, and regular checks on personal safety habits. The safety data sheets arrive thick and detailed, and every paragraph pays off when taken seriously.
Extra training always cuts risk. Supervisors who spend time reviewing emergency procedures and rehearsing spill response routines set their labs up for fewer incidents. Chemists often use less toxic substitutes when the job allows. More universities now track hazardous compounds with digital logs and barcodes, flagging old bottles before they leak trouble. Waste contractors work with labs to neutralize fluoride before final disposal.
Staying safe with tetraethylammonium fluoride trihydrate comes down to respect. Respect for the risks, respect for the science, and above all, respect for people—not just those in the lab, but anyone who could be affected down the line. Taking shortcuts cuts safety, and nobody wants to find out what that really means.
Countless researchers and industry folks ask about the molar mass of certain compounds without getting much beyond the textbook answer. For Tetraethylammonium Fluoride Trihydrate, a straight calculation points to 261.36 g/mol. Let’s break down why that number sticks and why it matters to those handling chemicals every day.
This compound builds off a Tetraethylammonium ion—four ethyl groups locked to a nitrogen. Fluoride tags along, plus three water molecules hang on as trihydrate. Every bit counts toward the mass.
Sum them up and the calculator lands at 203.25 plus 19.00 and the water, for 261.36 g/mol. That’s not a trivia nugget—it's what most labs base their dosing and reagent prep on. Even experienced chemists double-check these figures because accuracy at the bench saves time and money.
Forget theory for a moment. In practice, weighing chemicals wrong means your reactions fizzle or give odd results. I remember following a poorly written protocol early in my career. The author missed water of crystallization—my expected yields tanked and troubleshooting cost a whole day. Trihydrates, like this one, demand attention or else experiments drift from repeatability.
A lot of beginners rush past hydrate terms in names, thinking they don’t make much difference. If you run syntheses or prep solutions, the real compound on the shelf often isn’t just the “pure” salt—it walks with those attached water molecules. I’ve seen new hires toss out calculations just guesstimating amounts, burning through valuable material and sometimes risking safety.
Most suppliers give a purity range, but small print sometimes shifts—one batch could have a hint more water. Labs that run tight processes—battery research, pharmaceuticals, synthetic chemistry—keep tabs by reweighing and even doing Karl Fischer titrations to check water. Over years, I’ve learned never to trust a label without secondary confirmation if the application is high-stakes.
Taking a couple extra minutes to double-check a chemical’s formula and hydrate state pays for itself. Modern digital databases and basic spreadsheets can flag errors before glassware ever comes out. Teams with strong habits document the form of every compound, keep old datasheets, and list lot numbers right in the notebook.
Science doesn’t always forgive shortcuts. That molar mass—261.36 g/mol—is more than arithmetic; it anchors reproducible science. With the right numbers, you not only hit your targets but also minimize waste and avoid surprises.