The story of tetrapropylammonium chloride goes back to the early days of quaternary ammonium compounds in the twentieth century. Chemists pushed boundaries with new alkyl ammonium salts, looking for better surfactants and phase transfer catalysts. Tetrapropylammonium chloride emerged from this experimental environment. Researchers in the 1950s started to test its properties for organic synthesis, especially as they sought alternatives to more caustic alkali metal catalysts. Over decades, the compound found its place not just in chemical reactions but also in studies of zeolite synthesis and advanced material chemistry. Its development mirrors the constant push-and-pull between chemistry for industry and curiosity-driven research.
Tetrapropylammonium chloride stands out for being both practical and adaptable. Used mostly as a phase transfer catalyst, it supports reactions where water-loving and oil-loving chemicals need to meet. In the lab, it acts as a building block for other more complex molecules. What surprises many is how often it turns up behind the scenes in manufacturing processes, especially when conditions call for steering clear of metal ions. In industries where purity counts, tetrapropylammonium chloride helps keep reactions running clean, cutting out unwanted byproducts that can spoil the final result.
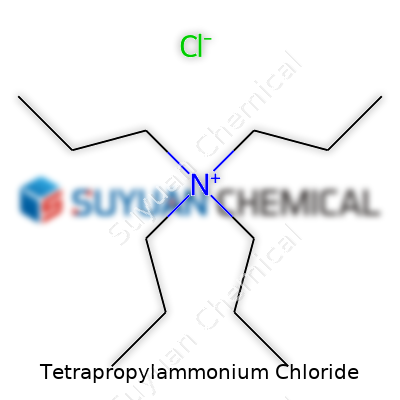
This compound usually shows up as a colorless or slightly off-white solid, easily soluble in water and a wide range of organic solvents. Its melting point tends to hover around 242–246°C, and it carries a molecular weight just over 229 grams per mole. These physical numbers only tell part of the story, though. The molecular structure—a nitrogen center surrounded by four propyl groups and countered by a chloride anion—gives it some fat, flexible organic arms, which let it play conductor between aqueous and organic phases. It shows strong thermal stability for an organic salt, and its ionic character means it can slip into polar solvents with ease, making it valuable for controlling reaction conditions.
Producers label tetrapropylammonium chloride with clear concentration and purity marks, often indicating purity of 98% or better for research and high-tech uses. Labels mention molecular formula (C12H28ClN), batch number, storage advice, and hazard warnings. Its CAS number—136-91-2—helps labs keep track of chemical inventories and regulatory reports. Handling guidelines stress use of gloves and goggles, since the compound can irritate skin and eyes even at the bench scale. While it doesn’t release choking fumes, it needs a sealed, dry container in storage—moisture can turn a clean white powder into a caked mess, complicating both measurement and reaction outcomes.
Industrial and lab processes tend to make tetrapropylammonium chloride by treating propylamine with propyl halide and going through a quaternization step. This approach lets chemists control yields by adjusting reaction temperature and solvent choice. The process spits out a crude product and a byproduct, generally an inorganic salt, which needs careful removal. Final purification usually involves recrystallization from ethanol or acetone, leaving behind a material that’s ready for prime-time applications. Over the years, improvements in batch process controls have boosted both yield and quality, avoiding trace contamination that can wreak havoc downstream.
Tetrapropylammonium chloride won’t steal headlines for wild reactivity, but its reliability in moving ions and molecules between phases gives chemists a way to unleash reactions that can’t happen in a single solvent system. It speeds up nucleophilic substitutions and organic oxidations by smoothing out mismatches between reactants—think of it as a backstage coordinator, not a lead actor. Over time, scientists have used it to shape zeolites, build ionic liquids, and even test ideas in molecular encapsulation. By swapping out the chloride for other anions, labs push into new territory, tailoring its properties to fit all sorts of reactions.
This chemical often answers to a handful of aliases, so knowing them cuts down on confusion. Common synonyms include TPAC and TPACl, along with trade names that might change between suppliers. Its systematic name—N,N,N-Tripropylpropan-1-aminium chloride—doesn’t roll off the tongue, but it keeps things clear in the regulatory world. Chemists experience confusion when scanning catalogs or safety data sheets, but a check of the molecular formula or CAS number usually clears things up.
Safety with tetrapropylammonium chloride means respect more than outright alarm. Short-term skin and eye contact causes irritation, so proper lab hygiene matters. Good ventilation, gloves, and goggles should always be part of the routine. Even though it’s not flammable, the compound puts off small amounts of dangerous gases if it gets too hot or ends up in contact with strong oxidizers. Operating standards in the chemical industry call for sealed handling systems and careful waste collection, since chronic exposure to quaternary ammonium salts can cause respiratory trouble among workers. Proper training prevents accidents, especially when moving between research labs and larger production lines.
Its real power lies in versatility. In organic chemistry, tetrapropylammonium chloride opens up new pathways for synthesizing esters, ethers, and other building blocks. In material science, it enables careful templating of zeolites—those tiny, porous crystals that clean up waste streams, refine oil, and even churn out new pharmaceuticals. Water treatment engineers rely on it for surfactant duties, stepping in where metals aren’t welcome. The electronics sector uses it to shape polymers and specialty glass coatings, especially when trace contamination threatens yields or performance. I’ve seen researchers rely on this compound as a dependable workhorse, lending a hand wherever selectivity and purity raise the bar.
Academic and industrial scientists continue to search for ways to get more out of tetrapropylammonium chloride. Development work focuses on swapping its chloride anion for others, making ionic liquids that resist water or carry out tougher catalysis. New reaction designs call for milder, more sustainable chemistry, and the compound’s flexible structure fits the bill. Researchers are looking at minimizing toxic waste and lowering the carbon footprint of phase transfer catalysis. In green chemistry projects, tetrapropylammonium chloride acts as a launching pad, introducing new partners in reaction schemes that skip hazardous reagents or cut energy use. Comparison tests between it and related ammonium salts reveal trends that guide better selection process for each recipe.
Scientists have put this compound through rigorous toxicology tests in recent years, seeking data relevant to both human health and environmental impact. Short-term contact usually causes just mild skin or eye discomfort, but longer exposure at high concentrations links to problems with respiration in some animal studies. Wastewater analysis detects the salt in low concentrations downstream from industrial plants—prompting closer checks for persistence and breakdown in the environment. Recent studies suggest that while it doesn’t build up in animal tissue like some heavier industrial pollutants, it can stunt aquatic organism growth at higher doses. This research pushes for improved effluent treatment and supports stricter workplace air and water monitoring.
Looking ahead, the role of tetrapropylammonium chloride will likely expand in green chemistry and sustainable manufacturing. Scientists are probing hybrid materials built on its backbone, aiming for next-generation catalysts and safer, cleaner synthesis of pharmaceuticals and polymers. With advances in process design, the focus is moving to reduce waste, cut energy use, and squeeze even more control over chemical selectivity. Future research trends point toward using this compound in precision medicine, biomaterials, and advanced nanotechnology. Changes in environmental standards will motivate cleaner disposal processes and push for safer alternatives where possible, but the lessons learned from decades of study will guide innovation rather than see this workhorse retired.
Tetrapropylammonium chloride doesn't show up in most homes or on grocery store shelves, but it does make a quiet impact in the background of chemical manufacturing. It's got a long name, but this chemical has a very targeted set of roles that help drive breakthroughs, especially in specialized labs and industries.
In a lot of organic chemistry research, this compound behaves like a traffic cop for ions—helping them move between water and organic layers in a reaction. Chemists call it a "phase-transfer catalyst." The appeal here is pretty direct. When trying to make certain molecules, sometimes the ingredients don’t naturally want to mix. Tetrapropylammonium chloride steps in and helps blend oil-and-water situations, opening doors for cleaner, more efficient chemical reactions.
And this is not a small niche. Cleaner processes create fewer byproducts, which shrinks factory waste and lowers disposal costs. That matters in everything from drugs to plastics. According to journals like Green Chemistry, using phase-transfer catalysts slashes unwanted side products by as much as fifty percent compared to traditional methods.
Beyond lab glassware, tetrapropylammonium chloride stands out for making zeolites. For anyone who’s not an industrial chemist, zeolites act like high-tech sponges at a molecular level. They clean up fuels and treat wastewater. Getting these zeolites just right often calls for a template to organize the inner structure like scaffolding on a high-rise. Tetrapropylammonium chloride lays down that blueprint, which leads to zeolites with pore sizes and shapes well-tuned for the job, whether that’s catching pollutants or separating valuable gases. Industries in petrochemicals and environmental engineering both rely on this process.
Safety doesn’t take a back seat with this substance. Like many ammonium compounds, it can be an irritant in concentrated form. Workers mixing or stirring it deal with gloves and goggles, plus good ventilation. These safety habits don’t just protect people—they help keep the worksite running without interruptions. The European Chemicals Agency requires detailed risk assessments for any large-scale handling, helping limit spills and accidental exposure.
As the chemical industry talks about cleaner and greener methods, phase-transfer catalysts like tetrapropylammonium chloride keep showing up in new approaches. Swapping out more toxic or energy-hungry catalysts for this one can deliver safer products and smaller carbon footprints. Research from the Royal Society of Chemistry points out gains in efficiency in pharmaceuticals and agrochemicals using these catalysts. If the aim is less waste and lower emissions, manufacturers gain extra incentive to retool old processes. More research into recycling and safer substitutes could widen options further, but for now, this compound still pulls its weight.
Most folks won’t ever buy a jug of tetrapropylammonium chloride, but its fingerprints touch everyday life through cleaner fuels, safer plastics, and healthier water. Progress in chemistry happens one smart tweak at a time, and this compound shows how even low-profile materials can drive the bigger changes that matter.
Tetrapropylammonium chloride pops up in a lot of research labs and industry settings. As someone who has handled chemicals for years, I’ve learned early that not all complicated names mean danger, but they always deserve respect. So what’s the real story about tetrapropylammonium chloride? Is it hazardous, or just misunderstood?
This salt shows up as a white, crystalline powder. It dissolves easily in water and helps researchers with everything from catalysis to the making of nanoparticles. The structure doesn’t scream “toxin” right away, but it pays to dig a little deeper and see what credible science says.
If you comb through reliable sources like the European Chemicals Agency and PubChem, you’ll find warnings around irritation. Skin, eye, and respiratory exposure bring risk of inflammation and discomfort, especially at concentrated levels. Most safety sheets urge the use of gloves and eyewear. In my experience, any compound that burns on contact deserves this basic level of protection.
Acute toxicity data stays pretty limited, but oral and inhalation routes can both produce harmful effects. Lab animal studies show possible impacts on the nervous system at higher doses, though there’s no direct link to cancer or heritable changes. That’s not a green light to slack off. With so many chemicals, we discover risks through long-term observation. Take it from someone who has seen workplace stories go sideways; those early “harmless” reputations fade quickly if safety steps are missed or ignored.
It also gets slippery if you’re in a setting with poor ventilation. Breathing in dust or vapor can make the throat burn and cause headaches or dizziness. As concentration increases, so does risk. Spills and splashes raise real concerns, too—accidents happen even to careful hands.
Waterways and soil can trap chemicals like this, especially when they’re used on an industrial scale. The Environmental Protection Agency notes that quaternary ammonium compounds have shown toxicity toward aquatic life. Tetrapropylammonium chloride does not flush through wastewater treatment unnoticed. These facts mean proper disposal matters as much as safe handling. In the past, careless disposal of other ammoniums led to real ecological damage.
Practical steps make a difference. In every research setting I’ve joined, smart habits saved more time than shortcuts ever did. Gloves, face shields, and lab coats should be standard. Anyone handling this compound should work in a fume hood whenever possible and avoid touching face or skin. If a spill happens, immediate cleanup and disposal in line with hazardous waste protocols stop far bigger troubles down the line.
In short, tetrapropylammonium chloride isn't as infamous as some toxins, but it's definitely not something to take lightly. Underestimating it, or ignoring established safety procedures, invites unnecessary health and environmental risks that can outweigh its scientific benefits.
Tetrapropylammonium chloride sounds like something you’d hear tossed around in a lab, but it’s pretty straightforward once you pull it apart. This compound features a core ammonium ion, with four propyl groups attached, and a chloride ion by its side. The chemical formula is C12H28NCl. That means every molecule includes twelve carbon atoms, twenty-eight hydrogens, one nitrogen, and one chlorine atom.
My first real encounter with tetrapropylammonium chloride was in a research setting that focused on phase-transfer catalysts. This compound gets used a lot for shuttling ions between water and organic solvents. That’s a lot more exciting in practice than it sounds on paper because it opens doors for otherwise impossible chemical reactions. Chemists turn to it when regular salts just won’t cut it. Its large, bulky structure lets it slip between layers in a reaction and promote mixing where other agents just hang back ineffectively.
If we look beyond the lab, tetrapropylammonium chloride’s formula isn't just trivia; it tells you what the molecule can do and why. The four propyl arms hanging off that nitrogen give it weight, but also allow it to dissolve in both water and organic environments. Those dual citizenship skills make it a key player when working in “two worlds" at once: the oily and the watery. Here’s where the formula gives away its secret — many quaternary ammonium salts have amphiphilic properties. The large, non-polar propyl groups and the positively-charged nitrogen mean that this molecule sticks its neck out and interacts with both greasy and polar molecules. That flexibility matters to anyone working on catalysis, drug delivery trials, or studying new materials that need a little coaxing to mix.
Getting a formula right saves time, money, and even health. I’ve seen labs waste expensive reagents because someone wrote down the name instead of the formula and grabbed the wrong bottle. Tetrapropylammonium chloride isn’t sodium chloride — a single atom out of place can flip a reaction’s outcome, ruin data, or put a halt to promising new tech. The numbers in the formula, twelve carbons and twenty-eight hydrogens, give every researcher and manufacturer a precise recipe to follow. No one needs a guess when measuring out compounds that might wind up in pharmaceuticals or sensitive electronics.
Chemistry can overwhelm newcomers, but information gets easier to tackle when backed by clear formulas. If a supplier shares only a name without a formula, I get skeptical quickly. Reputable companies don’t just list chemicals; they show structures, purity, and detailed specs. That kind of transparency builds trust, keeps processes reproducible, and supports regulatory standards. Mistakes, even small ones, can erode public faith. It’s always better to lead with clarity.
The chemical formula for tetrapropylammonium chloride guides safe handling. If someone thinks this compound behaves like regular table salt, they end up underestimating both its uses and risks. Training that spotlights accurate formulas, storage rules, and label reading helps workers avoid surprises in the lab or plant. Digital inventory systems, cross-checked by both name and formula, pull double duty: keeping research honest and ensuring workplace safety. Fact-based habits in chemical handling ripple outward, strengthening entire industries and protecting communities.
Tetrapropylammonium chloride appears in research labs, chemical supply rooms, and certain manufacturing processes. There’s a catch: this compound brings real safety issues. Many people notice its powdery look and clear crystal form and assume it acts harmless. That’s misleading. Touching or breathing this chemical invites skin and eye irritation, respiratory trouble, or worse. Flammable materials nearby can spell disaster if storage gets sloppy. In my work with chemical handling, I’ve seen accidents spark not from the substance itself, but from where and how someone leaves a bottle on a shelf.
Store tetrapropylammonium chloride in a cool, dry place. Humidity does more than cake up powders; it triggers reactions over time. Moisture creeping into containers leaves users with compromised chemicals and unexpected hazards. Keep this compound far from steam lines, sinks, windows, and the classic spot above a heater. Air conditioning and a low-humidity environment cut down on risks caused by water mixing in.
Leaving containers exposed to strong sunlight breaks down ingredients slowly. I’ve witnessed powders like this shift color and texture after months near a window. Those changes don’t just affect strength—they may create breakdown products that haven’t been tested for safety. Storage should always happen in a shaded cabinet or cupboard away from the sun. No one wants to find out what years of sun exposure did to the chemical’s properties.
Manufacturers package chemicals in containers designed to protect from air, light, and cross-contamination. Transferring the powder or solution into unlabeled jars or open bags looks tempting during busy times. In my experience, every shortcut with storage ends up costing more time through cleanup, risk reporting, and wasted product. Labels with chemical name, hazard symbols, and date received give everyone a heads up. Tight-fitting seals matter—this is one area where generic glass jars or food containers fail fast.
Tetrapropylammonium chloride reacts badly with strong oxidizers. Nearby bottles of bleach or peroxide raise the odds of violent chemical reactions. In one warehouse I visited, staff kept everything with an “A” in the name on a single shelf. The naming trick backfired as soon as they mixed a flammable liquid with an oxidizer, calling the fire department (and OSHA) within minutes. Segregate this chemical by its hazard class, not its spelling. Flammables, acids, and bases deserve their own zones.
Workers often skip reviewing safety sheets, trusting to memory. From my own days training technicians, I wish safety information hung near every shelf. Everyone in the space—from part-time lab assistants to full-time chemists—should wear gloves, goggles, and a dust mask at minimum. Pouring or mixing even a small amount in an unventilated space leaves people with headaches or rashes within hours. A chemical can stay stable for years with steady temperatures, sealed containers, and respect for its hazards.
If organizations make these habits routine, emergencies grow rare and everyone goes home safe. Basic respect for chemical properties—a lesson that matters whether you’re a student or a seasoned professional—pays off every time.
Take a pinch of Tetrapropylammonium Chloride (TPACl) and mix it into water. You watch as the white crystals dissolve, almost eager to vanish into the liquid. The salt, with its large organic cation and small chloride anion, interacts easily with water, thanks to the ability of water molecules to surround and stabilize its ions. I’ve worked in a handful of labs where TPACl sat on the shelf for months, and the instruction was always the same: measure it carefully, since it’s quite soluble around room temperature.
Unlike sodium or potassium salts, which nearly disappear in water instantly, TPACl’s chunky propyl chains make it slightly less zippy in dissolving, but it still dissolves well enough for typical benchwork. Most published sources pin its water solubility at over 50g per 100 mL—far more than needed for standard synthesis or analysis.
Move into the realm of organic solvents, and you see a drop in solubility. TPACl does better in polar environments, so solvents like methanol and ethanol hold onto the salt, though the numbers fall compared to water. Taking acetone or acetonitrile, TPACl still dissolves but nowhere near the extent in water. Try adding it to ether or hexane, the picture changes entirely—now TPACl settles at the bottom, refusing to interact with nonpolar surroundings.
Researchers and chemists have exploited this selective solubility for decades. Let’s say you want to separate out TPACl from a reaction mixture or do a cleanup step after synthesizing a product. Sometimes you’ll use an organic wash, knowing the bulk of TPACl will stick with the aqueous layer, leaving less residue in the final purified material.
TPACl’s solubility quirks shape its role in chemistry labs. It serves as a phase transfer catalyst, shuttling ions between aqueous and organic layers without fuss. The fact that it enjoys water more than most organics means it helps bring together substances that don’t want to mix. This feature speeds up reactions, especially for those chasing efficiency or aiming to scale up production without extensive purification steps.
Environmental health and safety play a part too. Since TPACl is so water-friendly, cleanup often involves simple aqueous rinses, which cuts down on harsh solvent use and costs. On the other hand, its limited solubility in nonpolar solvents minimizes the risk of accidental contamination in organic waste. Still, you shouldn’t pour TPACl down the sink; proper waste disposal matters, as with all ammonium salts.
One recurring challenge comes from the balance between solubility and selectivity. In chromatography, TPACl sometimes disrupts the stationary phase if dissolved in excess. Careful measurement, pre-dissolving in water, and slow addition to mixtures can sidestep this issue. I once watched a colleague foul up a silica column by overdosing TPACl—lesson learned, always check compatibility between your salt and your technique.
Tuning solubility to your benefit means choosing the right solvent and working within the limits of TPACl’s characteristics. With thoughtful use, TPACl helps bridge gaps in solubility, making tough reactions easier and cleanup less tedious. Knowing where and how it dissolves saves both time and resources on the bench and in production.