Triethylamine hydrochloride didn’t spring into labs overnight—chemists started working with its parent, triethylamine, back when amines shaped the backbone of early synthetic work in both Europe and North America in the early twentieth century. Chemical texts from that time already mention making salts to study reactivity and improve handling. Triethylamine itself showed up as a strong base around the time dye chemistry became a driving force for industrial progress, and making its hydrochloride salt turned out to be a practical way to store, ship, and dose this otherwise pungent, unstable, and volatile liquid. Established producers such as BASF, Merck, and others in the chemical boom years moved quickly to offer pure, stable salt forms by the 1940s, since researchers wanted materials they could handle in open air and dose by weight with less eye-watering risk. Watching industry shift toward robust, storable intermediates like this tells a story of improving both economy and safety in every generation of chemical manufacturing.
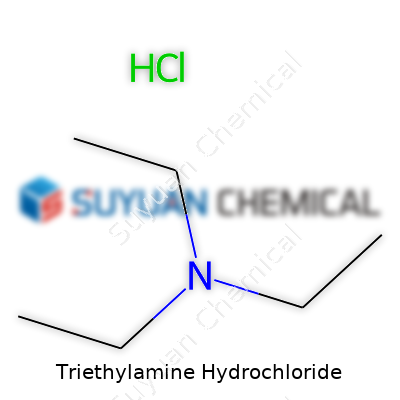
Triethylamine hydrochloride, cataloged in chemical supply houses under a scattering of names, pops up as a crystalline powder with a faint odor, packing the strong basicity of its parent within a solid that labs can weigh and mix with fewer headaches. This salt forms when triethylamine gas or liquid meets hydrochloric acid, producing a white, often fine powder that finds its way into pharmaceuticals, organic synthesis, and teaching labs. Purchasing agents see a range of packaging: sealed drums, foil pouches, and high-purity bottles marked by rigorous batch identifiers and shelf-life dates. The solid’s main selling point is its stability, offering a more user-friendly path for precise acid-base reactions or as a template in organic routes needing a non-nucleophilic base source. For relatively little cost, users can scale from gram samples to multi-kilogram quantities that flow into everything from pilot plants to undergraduate procedures.
Triethylamine hydrochloride stacks up as a solid at room temperature, dissolving well in water and lower alcohols. Its melting point clusters above 250°C, though with decomposition, so it doesn’t behave like salts that just liquefy and pool. Testing with common indicators reveals solutions with a neutral to slightly acidic pH, reflecting the full protonation of the amine by HCl. The white crystalline form may turn clumpy in humid conditions, absorbing moisture but not releasing much vapor, so operators breathe easier around the salt compared to the amine gas. Odor lingers, but nowhere near the eye-watering strength of free triethylamine. It weighs in with the formula C6H16NCl, clocking a molar mass just under 137.7 grams per mole. Anyone used to handling bulk salts soon learns that simple storage below 30°C in low-humidity cabinets helps avoid caking that could complicate dosing or mixing.
Suppliers present triethylamine hydrochloride under CAS Number 554-68-7, often with purity ratings touching 98% to 99.5% for laboratory use. Labels feature clearly indexed storage conditions (cool, dry), hazard markers, and batch traceability, with some offering certificates of analysis and lot-specific impurity profiles. Routine lot testing covers melting range, pH of a 10% solution, and residue on evaporation. Regulations in the US and EU set permissible levels of common heavy metal contaminants—lead, mercury, arsenic—well below a few parts per million. Shipping labels cite GHS hazard warnings, usually focusing on respiratory and eye irritation, and the need for gloves, goggles, and controlled ventilation during use and handling.
On the lab bench, making triethylamine hydrochloride looks straightforward: you add hydrochloric acid to triethylamine under cooling, keeping the exotherm under control. The reaction runs to completion almost as soon as the acid hits the base, yielding a slurry of white crystals. After stirring and cooling, filtration gathers the salt, which then dries under vacuum or in drying ovens. Production at scale tweaks the order of addition, sometimes sparging gaseous HCl into a solution of triethylamine in ethanol or water, favoring controlled nucleation and crystal size that suits either industrial large-reactor runs or smaller bottle-filling operations.
Triethylamine hydrochloride gives up its chloride ion in water, and in organic synthesis, it plays host to metathesis reactions where the Cl− swaps out for other counterions. Treating the salt with bases like sodium hydroxide or potassium carbonate regenerates the free amine, a trick many chemists use to purify or dry triethylamine as needed. In fields like peptide chemistry, this salt offers a buffered amine environment for coupling protocols without drifting pH problems. Other modifications, such as selective deprotonation, take advantage of the predictable release of triethylamine base in situ, which means the salt packs a versatility that makes it a staple on the shelf.
Walking through catalogs, you’ll see triethylamine hydrochloride listed under synonyms like N,N-diethylethanamine hydrochloride, TEA·HCl, and ethane, 1,1,1-triethylamine hydrochloride. European suppliers may favor “hydrochloride de triethylamine” or similar translations, and drug registries pitch terms like Acetyldimethylamine hydrochloride for regulatory filings. It pays to scan Safety Data Sheets carefully, as cross-references among synonyms ensure all handlers know what’s in the bottle—no matter what language shows up on the label.
Safety lessons with triethylamine hydrochloride start with basic personal protective equipment: nitrile gloves, eye protection, and fume hoods. While triethylamine itself triggers acute sensitivity in the airways, the salt version drops the risk profile considerably, though enough dust in the air can irritate eyes or throat. Spills on the bench clean up easily with damp towels or vacuum systems rated for fine powders. Disposal calls for standard salt solutions, checking local regulations for chloride release or high-organic content—industrial shops often funnel wastes through acid/base neutralization and certified incineration. Chronic toxicity rarely poses a problem, as long as operators respect exposure limits and don’t eat, drink, or smoke in work zones. Emergency response plans rarely need activation, but shop supervisors drill for accidental mixing with strong oxidizers or bases, since low-level byproducts could evolve irritating fumes.
Industries reach for triethylamine hydrochloride when seeking reliable, measured basicity in a solid dose form. Drug developers use the salt to deliver the triethylamine moiety without the volatility and storage headaches that plague the free base. In plastics, it helps as an intermediate during polymerization, especially where gradual basicity or chloride content calibrates reaction rates. Analytical chemists pick the salt for calibration standards or for generating reproducible pH environments in chromatography. University labs add it to undergraduate kits to demonstrate salt formation, buffer chemistry, or as a steppingstone to more elaborate peptide synthesis protocols. The pharmaceutical sector taps into its well-characterized impurity profile and shelf stability to standardize commercial library preparation, driving value beyond its relatively simple structure.
Discovery chemists gravitate toward triethylamine hydrochloride as a way to screen for reaction conditions in acid-sensitive transformations, where the parent base would skew yields or compete in side reactions. Instrument makers exploit its defined melting and solubility points for calibration and method validation studies. One growing research thread looks into modifying the salt itself—swapping chloride for less nucleophilic anions or preparing co-crystal analogs to tune reactivity or reduce handling hazards. In my own graduate school days, this salt proved vital during site-directed mutagenesis of nucleic acids that used base-catalyzed hydrolysis: packaged as a standardized, weighable solid, it allowed for protocol reproducibility from one year’s cohort of students to the next. Newer biotechnology fields now test blends of quaternary amine salts for cell-free production and gene editing, seeking improvements on decades-old practices built around staples like triethylamine hydrochloride.
Toxicologists collect data from acute and chronic studies of triethylamine hydrochloride to assess oral, inhalation, and dermal exposure risks. Historical studies roll back to basic LD50 values in rodents, suggesting relatively low oral toxicity but enough irritation that occupational limits matter. Researchers often focus on breakdown in living systems—tracking amine group metabolism and chloride excretion. Modern genetic toxicology screens show little sign of mutagenicity at concentrations below workplace safety thresholds, and long-term exposure tests suggest neither carcinogenicity nor reproductive toxicity emerges at levels typical for labs or industry. Still, routine risk reviews pop up as global regulations push further down the hazard and environmental persistence spectrum, especially where chloride runoff or amine traces persist in surface waters.
Triethylamine hydrochloride doesn’t fit the flashy profile of next-generation nanomaterials or high-energy polymers, but its future appears steady and robust as chemistry scales adapt toward safer, more sustainable platforms. Producers now crank up upstream traceability, delivering low-residue, low-carbony content grades for microelectronics and specialty formulations. Emerging green chemistry initiatives eye alternative counter-anions or demand less energy-intensive production cycles—here, closed-system reactors, solvent recycling, and smart packaging could lower environmental impact. On the synthetic front, custom co-crystals and salt forms based on triethylamine frameworks open new pathways in organocatalysis, ionic liquid development, and drug delivery. From where I sit, makers and users will keep the salt a perennial favorite, learning from decades of practice and continuing to push for higher precision, cleaner ports, and safer ways to harness this unassuming but crucial staple.
Walk into any chemistry lab, and you’ll spot all sorts of bottles and jars, each with a story. Among them, triethylamine hydrochloride tends to look pretty nondescript, but the work it enables goes far beyond its plain appearance. Most people won’t ever hear about it on the news, but this chemical drives progress behind the scenes in industries from medicine to electronics.
Triethylamine hydrochloride acts as a workhorse in organic synthesis. Chemists choose it for its ability to nudge molecules in the right direction. In pharmaceutical labs, it plays a key role by helping stitch together ingredients that end up in prescription drugs. Without compounds like this, everyday pills for conditions such as high blood pressure, depression, or allergies would be harder—sometimes impossible—to manufacture in a reliable way.
My first time working with triethylamine hydrochloride, I remember measuring out the precise amount for a reaction. The caution on the label stood out: treat with respect, measure carefully. Every chemist learns quickly that using too much or too little can derail months of planning. It functions as an efficient acid scavenger or as a catalyst, steering reactions so that only the desired product emerges with minimal waste. That means drug makers can deliver medications that do their job without impurities hitchhiking along for the ride.
This compound finds its place in other industries, too. For example, in materials science, it helps to prepare polymers and resins, those tough, versatile plastics in everything from coating materials to electronics. In electronics, it helps treat the surfaces of circuit boards to make sure they stay reliable even after years in a computer or a car dashboard exposed to sunlight and heat.
It’s also a favorite for creating specialty dyes and pigments, especially the ones that give high-end fabrics or inks their vibrant, lasting color. The reliability that triethylamine hydrochloride brings to dyeing processes shows up in everything from lab coats to high-energy athletic wear where colorfastness matters over repeated washes.
No one learns chemistry in a vacuum. A strict focus on safety rules always comes first. Triethylamine hydrochloride has some bite—it can irritate the skin and respiratory system if handled carelessly. That means wearing gloves, eye protection, and having good ventilation. After reading about a lab accident linked to poor ventilation, I always check for air flow before opening any bottle with a strong odor. Regular training and clear labeling prevent most incidents, and engineers are working to find ways to contain fumes more effectively.
Using chemicals like triethylamine hydrochloride raises important questions about health and environmental responsibility. Regulations require careful documentation, secure storage, and clear records about how much gets used and where leftovers go. Chemical waste disposal grows complicated, and companies often invest in treatment systems that neutralize byproducts before they reach the water supply. Researchers, including those in start-ups, have started pursuing greener alternatives to the classic methods—finding less hazardous catalysts or recycling used reagents. These shifts take years and plenty of patience, but they open the door to safer workplaces and a lighter ecological footprint.
As industries look for ways to shrink environmental costs, the next wave of chemists faces a challenge. They need reliable, proven substances like triethylamine hydrochloride, but they also need to think ahead—experimenting with approaches that replace hazardous components without sacrificing performance. My experience in the lab tells me change doesn’t come overnight, but every improvement counts. Asking tough questions, embracing new training, and adapting to new guidelines will make the biggest difference in the long run. These choices ripple out from the lab bench to the final product and then into our lives, reminding us that the smallest bottle in a lab often carries the biggest punch.
Triethylamine hydrochloride shows up in more labs and research spaces than most realize. If you’ve spent time near chemical shelves in a university, you probably spotted it among bottles lined up by the fume hood. Many researchers reach for triethylamine hydrochloride because it acts as a useful intermediate in organic synthesis. Still, comfort around such compounds sometimes leads to shortcuts. Chemists and even students, given the volume of lab safety training, have become pretty good at treating every bottle like it carries a threat.
Anyone who’s handled triethylamine itself recalls the smell—sharp and fishy, leaving a sting in the back of the throat if safety goggles slip for just a second. As a hydrochloride salt, this compound feels less aggressive, but the risks haven’t disappeared. Inhaling dust or fume, even from the hydrochloride form, irritates the nose, eyes, and throat; skin contact can cause itching or redness. The bigger worry emerges after repeated exposures or larger spills.
The chemical’s toxic profile stands out for its corrosive behavior. Workers in chemical manufacturing, where batches reach kilogram scale, face risk if they let guards down. NIOSH and OSHA flag triethylamine hydrochloride due to this corrosiveness and the potential for lung and skin irritation, urging vigilance even if safety data sheets limit themselves to terse warnings. Poisoning isn’t something most lab workers see every week, but a moment’s carelessness—the wrong gloves, a sprint for lunch before washing hands—brings the real danger into focus.
Experience says most chemical accidents link back to routine. Pouring a powder with no splash, tossing the weighing paper, or working late to finish a deadline—all can lead to forgetting a mask or skipping goggles. That’s when a simple compound turns into a skin or respiratory hazard.
Protective equipment plays a huge role. Proper nitrile gloves, splash-resistant goggles, face shields, and chemical-rated coat—these pieces might feel over the top, yet anyone burned or breathing heavily after inhaling powder knows their value. Chemical fume hoods take dust out of your face, making accidental inhalation less likely. Shortcuts get tempting after years of safe use, but chemicals don’t check if you’re experienced before causing burns or rashes.
Beyond individual vigilance, smart policies elevate the entire work environment. Training must emphasize real-world scenarios, not just slides or flashcards. Updating safety protocols after every incident—even minor ones—turns accidents into learning opportunities for everyone, not just the unlucky person involved. Facilities that supply clear signage, plenty of eye wash stations, and accessible safety showers earn trust from their workers. Keeping chemicals stored with desiccants and labeled in plain language makes haste less dangerous during busy days.
Routine audits and peer checks catch risks before they escalate. In smaller labs, researchers sharing mistakes openly lightens the pressure to play tough or silent. Safety culture doesn’t grow overnight, but clear leadership and soft reminders get everyone to treat even the familiar chemicals with the respect they deserve. Triethylamine hydrochloride doesn’t make headlines like cyanide or mercury, but underestimating it opens the door to preventable injury.
I remember working in a small research lab where we kept all our chemicals on mismatched shelves, just out of habit. Triethylamine hydrochloride sat next to acids and open bottles of solvents. Looking back, it’s clear that nobody gave much thought to how a single careless move could have led to disaster. There’s a tendency to treat certain reagents like they won’t cause harm until something actually goes wrong. That approach leads to ruined experiments or worse—injury and environmental fallout.
Triethylamine hydrochloride absorbs moisture out of the air, a property known as being hygroscopic. If it gets damp, you’re left with a sticky mess that is tough to weigh precisely and even tougher to work with in sensitive reactions. Leaving the cap loose or shoving the bottle in a cluttered cabinet might seem harmless at first, but over time, the compound loses reliability. Those of us who’ve found hard clumps in bottles mid-experiment know how frustrating that gets when research time and money are on the line.
This isn’t only about keeping out water. Fumes from strong acids or oxidizers can seep into neglected bottles—sometimes causing the material to react or degrade. I learned quickly that using airtight, chemical-resistant containers protects both the chemical and the people around it. Glass or high-density polyethylene bottles with sturdy seals do the job. Stick to labels that can't smear or fall off, since wiping dust off mystery bottles is a gamble no one enjoys.
Cool, dry storage isn’t just a nice suggestion—heat speeds up chemical breakdown, and hot, cramped storage closets encourage dangerous fumes. At one major university, a poorly ventilated room filled with excess cardboard and heat-trapping equipment turned one minor spill into a lingering headache for the entire corridor. Room temperature in a clean, well-ventilated spot works for triethylamine hydrochloride. Don’t stack heavy items on top, or squeeze containers onto overcrowded shelves either. Overlooked containers can fall, break, or leak unnoticed for weeks.
No matter how many safety data sheets pile up in a binder, mistakes happen if people don’t know the basics. Proper training on handling and storage helped my teams avoid health issues related to accidental skin or eye contact with triethylamine hydrochloride. Gloves, goggles, and coats aren’t overkill here. Accurate records matter, too. Tracking use lets you avoid buying excess chemicals you can’t store safely. The Chemical Safety Board has documented enough preventable incidents in academic settings to drive home exactly how important this is for everyone’s wellbeing.
Leftover triethylamine hydrochloride doesn’t belong down the drain or in common trash cans. Waste handling rules ask for labeled, closed containers and disposal through licensed handlers. People sometimes ignore this if they think the volume is too small to matter, but over time, small bottles add up to real hazards for water supplies and sanitation workers. Universities and companies should have streamlined protocols for collecting and treating even minor chemical waste. This is the only way to protect people and the planet in the long run.
Triethylamine hydrochloride isn't a term that hits headlines, but in chemistry labs, it shows up often enough to deserve a second glance. Its chemical formula, C6H15N·HCl (or more simply, C6H16ClN), spells out its real identity: a salt formed when triethylamine meets hydrochloric acid. That simple act changes things. The transformation from a smelly, liquid base to a crystalline, easier-to-handle solid has real value in both research and industry.
I remember in early laboratory days, dealing with triethylamine brought tears to the eyes – the aroma stings and lingers. The hydrochloride version, though, sits as a solid, much less likely to evaporate and less aggressive on the nose. Chemists appreciate this shift, as it allows labs to store a functional amine safely. With the hydrochloride salt, handling and measuring become less hazardous, especially for new technicians. No need to dread accidental spills wafting powerful fumes around the workspace.
Triethylamine hydrochloride acts as a standby in the toolkit for organic synthesis. Its presence matters in reactions where a milder, less volatile amine source fits better. Over the years, I watched colleagues choose the salt over the base when working with moisture-sensitive compounds or planning to scale up a reaction. That stability offered by the hydrochloride salt holds significance when planning for reliability and reproducibility.
Looking at recent developments in pharmaceutical manufacturing, controlling purity matters more than ever. By using triethylamine hydrochloride, firms avoid excess base, limit side products, and hit tighter specifications. The hydrochloride salt helps simplify product isolation in certain syntheses, helping purify target molecules. Documented research has shown its use in peptide coupling and pharmaceutical intermediate preparation, underscoring why understanding its chemical composition helps with process planning.
Concerns around waste and chemical exposure keep tightening. Triethylamine alone, after an accidental spill, becomes a headache for the environmental health folks. Its hydrochloride salt, less volatile and water-soluble, presents a smaller risk for air contamination, making it easier to clean up and dispose of. This difference shows why details like "hydrochloride" matter in practice, not just on the label or in a notebook.
In the wider push for greener and safer laboratories, making thoughtful choices about reagents protects both workers and the environment. Triethylamine hydrochloride’s formula packs practical lessons. Chemistry doesn’t only happen in glassware; it transforms how we handle, store, and clean up materials. Thinking through the implications of a formula like C6H16ClN helps set policies that keep everyone healthier and support responsible science.
More labs could benefit from training that highlights the practical safety advantages tied to different chemical forms. Encouraging routine use of safer salt forms in teaching labs and small-scale production would make workplaces less stressful and a bit more welcoming for newcomers. Updated storage policies and better labeling can go a long way to reduce accidents involving volatile amines. Real progress in lab culture often follows from small switches, like picking the salt form over the base.
Triethylamine hydrochloride shows up in a bunch of labs and plants. It’s useful, but it’s no friend if you treat it carelessly. I remember my first few months working in research; nobody took shortcuts around this stuff. One whiff tells you it can sting your nose and eyes. Long sleeves and gloves weren’t just rules—they were lifesavers. Goggle fog and glove sweat become minor complaints compared to what happens if you’re reckless.
This chemical gives off toxic fumes if it gets too hot or if you mix it wrong. Some folks don’t realize that once you break open a container, that sharp smell means ventilation fans should run, and open flames are a no-go. It isn’t about paranoia; it’s about leaving work with your health intact. A lot of traditional lab guides sing the same tune, but spending a day with a persistent headache or raw throat because of a spill gives you a fast education.
Skin burns, eye redness, shortness of breath—these are more than textbook warnings. Once, a spill on a benchtop forced us to vacate for the rest of the afternoon. Even small errors can throw off the whole team.
The material belongs on a dedicated shelf—never just anywhere. It deserves a sealed, clearly labeled container. If you’ve seen folks stashing it beside basic cleaners or on the floor, that’s a red flag. A secure chemical cabinet with good ventilation keeps headaches away on a busy day. Experience has taught me: skip proper storage, and you’ll regret it.
Nitrile gloves get pulled on first, long before grabbing any bottle. Face shields take priority if fumes drift your way. Fume hoods hum for a reason. We always check the spill kit every week; the backup-eye-rinse bottle gets its own test cycle too. Working with chemicals like this never becomes routine, no matter how many times you weigh or mix it. Vigilance isn’t overkill; it’s common sense.
If a spill happens, no one ever pretends it didn’t. We get out, alert everyone, and let the right people handle cleanup. Most old timers will tell stories about someone who thought a little splash meant nothing, only to spend hours in the ER or on paperwork later.
Dumping waste down the sink sounds faster until you realize what lands in the water supply. Used up triethylamine hydrochloride calls for a chemical waste drum, labeled, logged, and sealed. Waste coordinators pick it up—not just anybody. Before I ever poured a drop, I had to log hours on waste forms. This level of accountability became a habit. We rely on environmental compliance officers because they keep our operations on the right side of the law, not to mention protecting the neighborhoods near our campus.
Plenty of places work with local authorities to keep disposal safe and legal. Sometimes solutions look high-tech, with neutralization or professional incineration. The method doesn’t matter much as long as it follows the rules and protects workers, waste handlers, and communities downstream.
Clear communication, consistent training, and updated protocols make a big difference. People get lazy about checklists, but nobody’s too busy to prevent a disaster. Young chemists or old hands, everyone benefits from the reminders: respect the hazards and put safety on autopilot. Triethylamine hydrochloride demands it, and so does anyone who values a safe workplace at the end of the day.